Introduction
Hamstring muscle injuries are prevalent among physically active individuals and athletes, particularly those participating in sports that require high-speed running, kicking, or sprinting. These injuries can substantially affect training capacity, match availability, and overall function.1 Modern soccer (football) is characterized by dynamic, intermittent patterns of play with repeated high-speed actions such as accelerating, jumping, and rapid changes of direction, all performed under considerable physical and technical demands.2
The hamstring muscle group consists of three major muscles located in the posterior aspect of the thigh: the semimembranosus (most medial), the semitendinosus (intermediate), and the biceps femoris, which has long and short heads (most lateral).3 Their biarticular structure, spanning both the hip and knee joints, together with the high loads generated during eccentric actions, contributes to their vulnerability to injury.4 Movements involving simultaneous hip flexion and knee extension, or rapid transitions between these positions, can place substantial strain on the hamstrings and increase the risk of muscle strain.4 Hamstring strain is among the most frequent injuries in sports medicine; it has been reported that hamstring injuries affect a large proportion of professional soccer players and account for a substantial percentage of all time-loss injuries.2
Epidemiological data from professional soccer and other high-level sports indicate that hamstring injury rates are high and that recurrence is common. A 10-year longitudinal study in the National Soccer League (NFL) reported an incidence of approximately 2.3 hamstring injuries per 1,000 athlete-exposures during practice, with hamstring injuries representing a substantial proportion of all recorded injuries.5,6 Typical recovery times range from roughly one to four weeks, depending on the location and severity of the injury.5–7 The incidence of hamstring injury is higher during matches than training, and time-loss varies with injury type, age, and seasonal period.5 These injuries therefore represent an important performance and health burden in elite soccer.
Randomized controlled trials have suggested that rehabilitation programs emphasizing progressive running drills and eccentric hamstring strengthening are associated with earlier return to sport and reduced reinjury rates.8–10 The primary goal of muscle injury rehabilitation is to restore normal neuromuscular function while minimizing fibrosis, scarring, and the risk of recurrence.11,12 In intermediate phases of recovery, athletes progressively regain functional strength and neuromuscular control through exercises of increasing complexity and load. Consequently, appropriate management strategies and prevention programs are essential to reduce reinjury and delayed return to play.12
Therapeutic ultrasound (TUS) has been widely used in the rehabilitation of musculoskeletal injuries. Its acoustic waves can induce mechanical and thermal effects at the tissue level and may alter cellular activity and ion gradients.13 TUS is relatively accessible and inexpensive and is frequently used as an adjunct modality in clinical practice.14 However, the overall evidence regarding the effectiveness and clinical relevance of TUS in soft-tissue injury management is mixed. Several systematic reviews and clinical practice guidelines have reported limited or no clear additional benefit of ultrasound on pain, function, or time to recovery for many musculoskeletal conditions, and some studies have not demonstrated superiority over control conditions. This uncertainty highlights the need for well-designed trials evaluating TUS as an adjunct to active rehabilitation, particularly in specific populations such as elite athletes.
The primary objective is to describe the protocol of a randomized controlled trial designed to assess the efficacy of a combined program of therapeutic exercises and TUS compared with therapeutic exercises alone for the rehabilitation of partially injured hamstring muscles in elite soccer players. The secondary objective is to examine the effect of the intervention on time to return to full team training and match eligibility (“quick recovery”), as well as on several physical and clinical outcomes, including hamstring muscle strength, electrical activity of the hamstrings, and pain perception. By focusing on a homogeneous group of elite soccer players and using clearly defined outcomes, this trial aims to contribute to the ongoing debate regarding the role of TUS as an adjunctive modality in contemporary sports rehabilitation.
METHODS
Study design
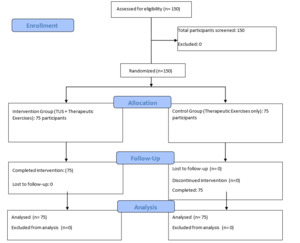
This study will be a single-center, parallel-group randomized controlled trial designed in accordance with the Consolidated Standards of Reporting Trials (CONSORT) guidelines15 and the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT).16 A flowchart of the study design and follow-up is presented in Figure 1. The trial will be conducted at a public university in the Middle East, within the College of Sports Sciences and Physical Education. Data collection will take place in university facilities, including the physiotherapy center, fitness and strength training hall, and track and field arena.
Participants
The study will involve elite male soccer players from eight professional soccer clubs competing in a national premier league. Players will be identified and contacted through the medical teams of the participating clubs.
Sample size
Based on data reported by Vermeulen et al.17 for days to recovery, and assuming an effect size of Cohen’s d = 0.75, a two-sided α < 0.05, and 80% power, it was calculated that 128 participants would be required. Allowing for an anticipated attrition rate of approximately 15%, the final target sample size for this trial is 150 participants in total (75 per group).
Recruitment procedure
Recruitment will commence after obtaining approval from the participating clubs to implement the intervention. Potential participants will receive detailed oral and written information about the study aims, procedures, potential benefits, and risks to support informed decision-making. Those who agree to participate will provide written informed consent before any study-specific assessments are conducted.
Collaboration will occur with soccer clubs and a sports medicine department as research partners. Eligibility criteria will be applied sequentially until the required sample size of 150 participants has been reached.
Eligibility criteria
Inclusion criteria
Eligible participants will be male players aged 18–35 years from the eight clubs in the Iraqi Soccer League who:
-
Are currently registered as elite players and regularly participate in team training and matches;
-
Report a sudden onset of posterior thigh pain during soccer training or matches consistent with a hamstring strain;
-
Present with clinical signs of partial hamstring strain (localized pain on palpation of the hamstring muscle and localized pain during at least one of the following tests: the Passive Knee Extension Test.
-
Have magnetic resonance imaging (MRI) confirmation of a partial hamstring injury.
Exclusion criteria
Players will be excluded if they:
-
Have other concomitant lower-limb or spinal conditions that could explain posterior thigh pain;
-
Have had a confirmed or suspected hamstring injury in the same leg within the previous six months;
-
Have general contraindications to MRI;
-
Are currently enrolled in another structured rehabilitation program targeting hamstring injuries.
Randomization and blinding
Participants will be allocated to the intervention or control group using a computer-generated random allocation sequence (Castor EDC software). An independent statistician will generate the sequence and conceal it using opaque, sealed envelopes. Baseline clinical data and informed consent will be obtained before randomization.
Because only the intervention group will receive therapeutic ultrasound, participants and treating therapists cannot be fully blinded to group allocation. To minimize bias, outcome assessors (those conducting strength, EMG, pain, and return-to-play assessments) and data analysts will remain blinded to group assignment. Opaque envelopes and separate scheduling procedures will be used to avoid accidental unblinding of assessors. Blinding adequacy for assessors and analysts will be evaluated using the James and Bang blinding indices.
Intervention
Participants will be randomly assigned to one of two groups: (1) Therapeutic Exercises and Ultrasound group (n = 75) or (2) Therapeutic Exercises-only group (n = 75). Both groups will attend three supervised sessions per week for six weeks. The intervention group will receive a combined program of therapeutic exercises plus therapeutic ultrasound, while the control group will receive the same exercise program without ultrasound (Table 1).
Therapeutic Exercises and Ultrasound program
Participants in this group will follow a standardized, phase-based rehabilitation program. The therapeutic exercises will focus on strengthening the muscles around the knee and pelvis, restoring neuromuscular control, and preparing the athlete to return safely to soccer-specific activities. The program will be applied individually, with progression tailored to each participant while maintaining a standardized structure.
Therapists will ensure that each exercise is clearly explained, demonstrated, and practiced with correct technique. Exercises will be graded from simple to more complex tasks, with attention to movement quality and symptom response. Participants will be encouraged to report pain or excessive tension, and unnecessary overloading or additional exercises outside the protocol will be avoided.
A daily session record (session transcript) will be completed for each participant. This record will document symptoms, clinical signs, and test results, including pain intensity, localized pain on palpation at the injury site, range of motion, muscle biopotential signal (EMG), and strength (see outcomes section for compete evaluation protocol).
During each supervised session, the participant will lie prone on a treatment table while the therapist applies therapeutic ultrasound to the injured region of the hamstring muscle. The Ultrasound sessions will be performed daily on the injured hamstring while the participant lies prone. Parameters will be: continuous duty cycle, frequency 3 MHz, intensity 1.0 W/cm², treatment head 5 cm², duration 5 minutes per session. These parameters were selected based on previous clinical protocols for acute soft-tissue injuries and manufacturer recommendations.
A standard coupling gel will be used, and a transducer head of approximately 5 cm² effective radiating area will be moved slowly over an area approximately 2–3 times the size of the transducer surface, the location of the application area of the head is determined by manually palpating the muscle belly with a pincer grasp to detect the area of maximum pain. These parameters are selected to target superficial soft tissue structures such as the hamstring muscle belly and proximal tendon.
The rehabilitation program will be divided into four phases: acute, subacute, remodeling, and functional (Appendix 1). Briefly:
-
Acute phase: focus on pain and swelling control, protection of the injured tissue, and maintenance of general mobility.
-
Subacute phase: introduction of gentle stretching and low-load strengthening exercises, such as towel stretch, slump stretch, standing hamstring stretch (for biceps femoris), and single-leg balance, supine two-leg bridge, and manual resisted hamstring exercise (for semitendinosus or semimembranosus injuries).
-
Remodeling phase: restoration of full range of motion and progressive strengthening, including exercises such as leg flexion and extension, isometric contractions at different angles, supine two-leg bridge, hamstring sets (heel dig at 30°, 60°, 90°), manual resisted hamstrings, upper limb strengthening, and cycling.
-
Functional phase: emphasis on advanced flexibility (e.g., PNF stretching of the hamstrings), resistance exercises (including Nordic hamstring exercises), and running drills with changes of direction (e.g., T-running exercises), aiming to prepare the athlete for pain-free, full-speed running and soccer-specific demands.
Progression from one phase to the next will be decided by the rehabilitation team based on predefined clinical and functional criteria, including pain levels, range of motion, and the ability to perform specific tasks without symptoms. This individualized but criterion-based approach is intended to maximize safety and optimize recovery (Appendix 1).
Exercise technique guidance
Detailed instructions will be provided for each therapeutic exercise. These instructions will be supported, when possible, by visual demonstrations (videos or in-person demonstrations) from qualified therapists (see subsequent sectin for qualifications). Emphasis will be placed on maintaining proper alignment, controlled movement, and appropriate muscle activation throughout each exercise. Therapists will provide ongoing feedback and corrections to ensure that participants perform exercises correctly and safely.
Progression criteria
Progression within and between phases will be guided by both subjective and objective indicators. Subjective indicators will include self-reported pain during and after exercises, perceived functional capacity, and perceived exertion. Objective indicators will include muscle strength assessments, range of motion measurements, and functional movement evaluations. Specific benchmarks, such as the ability to perform at least 10–15 repetitions of a prescribed exercise without pain or compensatory movement patterns, restoration of ≥90% strength symmetry compared with the contralateral limb, and the ability to complete basic functional tasks (e.g., jogging or controlled change-of-direction drills) without symptom exacerbation, will be used to guide progression. Participants will advance to the next phase only when these criteria are met, as determined by the treating therapists.
Therapeutic Exercises-only program
Participants in the Therapeutic Exercises-only group will perform the same phase-based exercise program as the Therapeutic Exercises and Ultrasound group, with the same frequency (three sessions per week for six weeks), duration per session, and progression criteria. The only difference will be the absence of therapeutic ultrasound. Session time and therapist contact will be matched as closely as possible between groups to minimize performance and attention bias (Table 1, Figure 2).
Therapist qualifications
The therapeutic exercises and ultrasound treatments will be administered by qualified therapists with expertise in musculoskeletal rehabilitation and sports medicine with at least two years of experience in treating soccer players. These therapists will hold relevant professional qualifications (e.g., physical therapists or athletic trainers) and will have experience working with athletes and individuals recovering from musculoskeletal injuries, particularly hamstring injuries. Therapists administering ultrasound will be trained in the safe and effective use of therapeutic ultrasound devices. Regular supervision and oversight by experienced rehabilitation specialists will help ensure that participants receive high-quality care consistent with current best practice guidelines.
Data collection procedure
Baseline questionnaire
At baseline, participants will complete a questionnaire to capture demographic and sport-related information. Variables will include body height and weight (measured using a SECA 225 stadiometer and scale; SECA, Hamburg, Germany), age, playing experience, anthropometric attributes, regular playing position, preferred kicking leg, level of competition, and history of hamstring muscle injury in the previous six months. Additional items will assess previous rehabilitation experiences, psychological readiness to return to play, and perceived risk of re-injury.
Magnetic resonance imaging (MRI)
MRI will be used to confirm and characterize hamstring injuries. Standard axial, coronal, and sagittal T1- and T2-weighted images will be obtained using a 1.5 T superconductive MRI unit (Magnetom Espree, Siemens, Erlangen, Germany). All players will undergo MRI within 5 days of the acute injury. A phased-array surface coil and two-body matrix coils will be positioned over the injured hamstring muscles while the participant lies supine.
MRI will be used to determine injury characteristics, including severity, involved muscles and tendons, and injury location. Images for proximal hamstring injuries will extend from the ischial tuberosities through the proximal thigh, and for distal injuries from the mid-thigh to the knee. Additional high-resolution images will be obtained through the injured thigh as needed. MRI will also be used to identify the precise site of injury (origin, proximal myotendinous junction, muscle belly, distal junction, or insertion), the number of involved tendons, the extent of retraction, and signs of chronicity.
Study outcomes
Baseline data
Baseline data will include participant characteristics (age, anthropometrics, playing position, preferred leg, competition level), history of hamstring injury and previous rehabilitation, and psychological readiness to return to play (RTP) and perceived risk of re-injury.
Primary outcome
The primary outcome will be time to return to play (RTP), defined as the number of days from the date of injury (day 0) to the date on which the player is medically cleared to fully participate in team training and be available for match selection.
Secondary outcomes
Medical clearance will be granted by the club medical staff based on the following predefined criteria:
-
No pain on palpation of the previously injured hamstring.
-
Pain-free passive and active range of motion (ROM) comparable to the contralateral side.
-
Restoration of hamstring strength to at least 90% of the contralateral limb, as assessed by isometric knee flexion testing (see outcomes section for compete evaluation protocol).
-
Symptom-free completion of the full rehabilitation program, including all functional tasks prescribed in the final phase.
-
Successful completion of a standardized running test (including high-speed running and changes of direction) without pain or functional limitation.
The date on which all five criteria are met and documented will be recorded as the RTP date.
Outcome variables will be:
Hamstring muscle strength
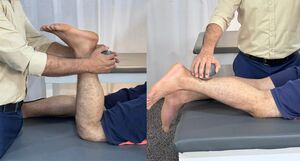
Hamstring muscle strength will be assessed using the ForHealth Kit (Chronojump Bosco System, Barcelona, Spain). Isometric knee flexion strength will be measured at two knee flexion angles (15° and 90°) in both the injured and uninjured legs, with the participant in a prone position.
For the 90° condition, the force sensor will be positioned horizontally against the posterior aspect of the athlete’s heel to measure isometric knee flexion strength at 90°. For the 15° condition, the force sensor will be placed vertically against the posterior heel to measure isometric strength at near-full extension. By assessing strength at both angles in both limbs, the study will characterize neuromuscular performance and potential strength asymmetries between the injured and uninjured legs (Figure 3).
Muscle biopotential signals
Surface electromyography (EMG) will be recorded during isometric contractions at 1000 Hz using the MyoTrace™ 400 system (Noraxon, Arizona, United States, Inc.). Self-adhesive silver chloride dual-snap electrodes will be placed over the hamstring muscles according to standard skin preparation and electrode placement guidelines. The MyoTrace 400 portable transmitter will send EMG data wirelessly via Bluetooth to a personal computer for storage and later analysis. This setup will allow remote recording of EMG signals from the target muscles during selected strength and functional tasks. Data will be analyzed as percentage of maximum voluntary contraction (MVC).
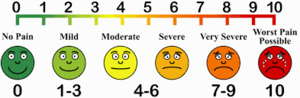
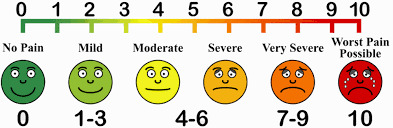
Degree of pain
Hamstring pain intensity will be assessed using a 100 mm visual analogue scale (VAS) (Figure 4). The VAS consists of a horizontal line anchored with “no pain” at the left end and “worst imaginable pain” at the right end. Participants will be asked to place a mark on the line corresponding to their perceived pain intensity at rest and during specific tasks. The distance in millimetres from the left end to the mark will be used as the pain score, with higher values indicating greater pain.
Monitoring
All medical teams from the participating clubs will be thoroughly briefed on study procedures prior to recruitment. Healthcare providers involved in the trial will receive initial training from the principal researcher regarding the study aims, procedures, outcome measures, and their specific roles. Practical workshops will be organized for local evaluators to practice the assessment procedures and the use of all equipment. Written manuals and standardized forms will also be provided. Study procedures will be monitored by the National Centre for Training and Human Development and by an independent monitor at the University of Baghdad.
Monitoring protocol
Participant progress will be monitored systematically at baseline and throughout each phase of the rehabilitation program with a evaluation at the end of each week during the six-week protocol duration. Regular assessments will be performed by therapists trained in the evaluation of musculoskeletal injuries. These assessments may include physical examination, functional movement assessments, and performance tests specific to hamstring function and injury recovery. Assessments will be scheduled at predefined time points and at transitions between phases to detect changes in condition and to inform progression decisions. Based on the assessment findings, the exercise program may be adjusted to optimize progress and address any emerging issues.
Adherence monitoring
Adherence to the exercise program will be documented across the study period. Participants will receive exercise logs or journals to record their participation in each supervised and, if applicable, home-based exercise session, including type of exercise, duration, and perceived intensity. Participants will also be encouraged to note any difficulties or barriers to adherence (e.g., pain, scheduling conflicts, lack of motivation). Therapists will review exercise logs during follow-up visits or remote consultations to evaluate adherence, reinforce compliance, and provide additional support or modifications when needed.
To explore the adequacy of blinding of outcome assessors and analysts, the James blinding index (values from 0 to 1, with higher values indicating better blinding) and the Bang blinding index (values from −1 to 1, with values between −0.2 and 0.2 suggesting adequate blinding) will be calculated at the end of the trial.
Statistical analysis
All statistical analyses will be performed using SPSS version 24.0 (SPSS Inc., Chicago, Illinois, USA). Analyses will follow the intention-to-treat principle, with all randomized participants included in the primary analysis according to their allocated group.
For the primary outcome (time to RTP in days), groups will be compared using survival analysis methods. Kaplan–Meier curves will be constructed, and the log-rank test will be used to compare time-to-event distributions between groups. In addition, a Cox proportional hazards model will be explored to estimate the hazard ratio for RTP between groups, adjusting for relevant baseline covariates (e.g., age, baseline injury severity) if appropriate.
For secondary outcomes, continuous variables (e.g., strength, EMG measures, VAS pain scores) will be summarized using means and standard deviations or medians and interquartile ranges, as appropriate. Between-group comparisons at baseline will be performed using independent-samples t-tests or Mann–Whitney U tests. To examine changes over time (e.g., baseline to post-intervention) between groups, two-factor mixed-model ANOVA or MANOVA will be used, with one between-subjects factor (group: Therapeutic Exercises and Ultrasound vs. Therapeutic Exercises-only) and one within-subjects factor (time: baseline and weekly (up to 6 time measuremetne times)). When assumptions of normality or sphericity are violated, appropriate corrections (e.g., Greenhouse–Geisser) or non-parametric alternatives will be applied. Post hoc tests with Bonferroni correction will be used to adjust for multiple comparisons when necessary.
Categorical variables (e.g., proportion of players experiencing re-injury during follow-up) will be compared between groups using χ² tests or Fisher’s exact tests. Levene’s test will be used to assess homogeneity of variances, and Mauchly’s test will be used to assess sphericity where applicable.
Spearman rank-order correlations will be used to explore associations between time to RTP and selected variables such as MRI injury characteristics and palpation findings. The significance level will be set at p < 0.05 for all analyses.
Ethics approval and informed consent
The study protocol was approved by the Ministry of Health / Research Committee of the National Centre for Training and Human Development. The trial is registered at ClinicalTrials.gov (NCT06165783). All participants will provide written informed consent before enrollment. Any important protocol modifications or amendments will be submitted to the Research Committee of the National Centre for Training and Human Development and reported in the final RCT report. The study will be conducted in accordance with the ethical principles of the Declaration of Helsinki (2008).
Discussion
This study protocol describes a randomized controlled trial designed to assess the efficacy of therapeutic exercises with or without therapeutic ultrasound for the rehabilitation of partially injured hamstring muscles in elite soccer players. The primary focus is on time to return to play, defined as the time required to recover sufficiently to fully participate in team training and be eligible for match selection according to predefined clinical and functional criteria. By prospectively evaluating this rehabilitation strategy in a homogeneous population of elite soccerers, the trial aims to provide scientific data that may assist medical staff and other healthcare providers in decision-making for the management of partial hamstring injuries.
Clinical predictors of return to play after hamstring injuries have been previously explored. For example, Marc et al. reported that athletes with chronic groin pain experienced less pain and improved sports performance after an active rehabilitation program emphasizing strength and coordination of the pelvic musculature, compared with programs focused primarily on passive modalities and stretching.18 Samuel et al. found that self-reported time to return to play and deficits in passive straight leg raise were significantly associated with time to return to play after hamstring injury.19 Warren et al. observed in a cohort of Australian soccer players that those who required more than one day to walk pain-free were more likely to need more than three weeks before returning to play.20 In a Danish study of elite and sub-elite soccer players with hamstring injuries, mean recovery times were approximately 30 days (range 7–64) and 26 days (range 4–89), respectively.21 These findings highlight the variability in recovery and the importance of identifying effective, evidence-based rehabilitation approaches that may reduce time loss and risk of reinjury.
Therapeutic ultrasound is frequently used in musculoskeletal rehabilitation; however, its effectiveness remains uncertain and somewhat controversial. Some experimental and clinical studies suggest that ultrasound can produce thermal and mechanical effects in soft tissues, potentially influencing pain, muscle spasm, and local circulation.20,22 Rantanen et al., for instance, reported that therapeutic ultrasound may induce vasodilation and modulate tissue temperature, which could theoretically support tissue healing.22 Other work, such as that by Wilkin et al., has examined ultrasound in relation to hamstring extensibility, with mixed findings and limited evidence for clear superiority over control conditions.13 Overall, the current literature presents inconsistent and often low-to-moderate quality evidence regarding clinically meaningful benefits of therapeutic ultrasound for acute soft-tissue injuries.
In this context, the present trial is explicitly designed to test therapeutic ultrasound as a potentially useful but not yet well-established adjunct to an exercise-based rehabilitation program. The authors hypothesize that adding therapeutic ultrasound to a structured therapeutic exercise program may reduce time to return to play and improve secondary outcomes (strength, muscle activation, and pain) compared with therapeutic exercises alone. At the same time, the authors acknowledge that the existing evidence base is mixed, and it is equally possible that no additional benefit will be observed. Regardless of the direction of the findings, the results of this trial should contribute valuable data to the ongoing debate about the role of therapeutic ultrasound in sports injury rehabilitation and may inform future clinical practice guidelines and research on hamstring injury management in elite soccer players.
This study protocol has several important limitations. First, although the trial is randomized, it is conducted at a single center and includes only male elite soccer players from one national league, which may limit the generalizability of the findings to other levels of play, other sports, female athletes, or non-elite populations. Second, the control group will not receive a sham ultrasound intervention. As a result, participants and treating therapists cannot be fully blinded to the use of ultrasound, and expectancy effects cannot be completely ruled out; we will, however, blind outcome assessors and data analysts and formally assess blinding adequacy. Third, the intervention protocol is relatively intensive and requires specialized equipment and experienced therapists, which may restrict its feasibility in some clinical settings. Finally, although clinically relevant outcomes and standardized criteria for return to play have been selected, unmeasured factors such as team selection policies, player motivation, and contextual match demands may influence the actual timing of return to competition. These limitations will be taken into account when interpreting the results and considering their implications for broader clinical practice.
Conclusions
This study protocol describes a rigorously designed randomized controlled trial evaluating the effects of a structured therapeutic exercise program with and without therapeutic ultrasound in the rehabilitation of partial hamstring injuries in elite soccer players. By focusing on clinically relevant outcomes such as time to return to play, muscle strength, neuromuscular activation, and pain, this trial aims to provide high-quality evidence to inform evidence-based decision-making in sports medicine and rehabilitation. The findings of this trial are expected to clarify the potential role of therapeutic ultrasound as an adjunct to exercise-based rehabilitation in elite athletes and to contribute to the development of optimized rehabilitation strategies for hamstring injuries in high-performance soccer.
Conflicts of Interest
The authors declare no conflicts of interest.
Acknowledgements
We wish to thank the Aragonese Primary Care Research Group (GAIAP, B21_23R) that is part of the Department of Innovation, Research and University at the Government of Aragón (Spain) and the Institute for Health Research Aragón (IIS Aragón).