INTRODUCTION
The Anterior cruciate ligament (ACL) is a key stabilizer of the knee joint, providing resistance against anterior tibial translation and rotational forces. It also contributes to proprioceptive control during high-speed and pivoting athletic activities.1,2 Biomechanically, the ACL plays a vital role in proprioception and load transfer during tasks involving cutting, deceleration, or rapid change of direction common actions in sports such as football, badminton, and basketball. The anterior cruciate ligament can rupture through contact or non-contact mechanisms, resulting in altered knee biomechanics, impaired neuromuscular control, and functional instability.3–6
Surgical reconstruction is widely accepted as the gold standard for restoring mechanical integrity, particularly in active individuals. However, the long-term success of ACL reconstruction depends more heavily on post-operative rehabilitation than on the surgical technique itself.7,8 Rehabilitation is designed to follow a phase-wise model beginning with pain control and range of motion, followed by progressive strengthening, proprioceptive training, and culminating in return-to-sport (RTS) activities.5,9 Despite the development of criterion-based RTS guidelines and neuromuscular training protocols, many subjects experience non-linear recovery, where expected improvements either stall or regress.
One often overlooked phenomenon in ACL rehabilitation is the rehabilitation plateau, defined as a phase in which physical, neuromuscular, or functional progress slows or stagnates despite adherence to a structured rehabilitation program.1 Although well described in neurological rehabilitation and other orthopaedic conditions, rehabilitation plateaus remain underrecognized and insufficiently explored in sports injury literature, particularly following ACL reconstruction.
In stroke recovery, plateaus are commonly described as points where motor or cognitive recovery halts, despite ongoing therapy.10,11 In orthopedic conditions such as total knee arthroplasty, recovery trajectories demonstrate a predictable slowing of range-of-motion gains beyond the early postoperative period.12,13 While this reflects an expected procedural recovery ceiling rather than pathological stagnation, it provides a useful framework for understanding how biological and neuromotor adaptation limits may also influence recovery pacing in ACL rehabilitation. These observations suggest that rehabilitation plateaus may be influenced by neuroplastic adaptation limits, persistent neuromuscular inhibition, soft tissue adaptation thresholds, psychological fatigue, and insufficient progression of therapeutic loading—mechanisms that have been widely described in neurorehabilitation and neuromuscular control literature and are increasingly recognized as relevant to ACL rehabilitation.1–4
These plateaus may emerge during critical transition points, such as moving from isolated strength to explosive single-leg loading or from basic drills to sport-specific reactivity. Biologically, this may relate to persistent quadriceps inhibition, altered cortico-motor excitability, or compensatory movement strategies.14,15 Psychologically, fear of reinjury, low confidence, or mental burnout may limit load acceptance.16,17 Moreover, external factors such as inconsistent rehabilitation attendance, poor therapist communication, and inadequate overload progression can further increase the likelihood of plateau onset18–20 and failure to integrate neurocognitive strategies21 can all lead to extended recovery periods and plateaus in both youth and adult populations.
Plateaus may occur at any phase of rehabilitation but are particularly evident during the transition from mid-stage recovery—focused on strength and motor control—to advanced sport-specific loading.22,23 During this phase, increasing physical demands require not only adequate muscular capacity but also precise neuromuscular coordination and load tolerance. When these requirements are not met, athletes may demonstrate delayed progression, persistent asymmetries, or difficulty advancing to higher-level functional and sport-specific tasks.
In rehabilitation science, plateaus are not necessarily permanent but represent an adaptive ceiling based on current stimuli. Breaking through this ceiling often requires a shift in strategy from traditional strengthening to cognitive-motor integration, sensory-perceptual retraining, and contextual movement planning.1,24 Recognizing and responding to these plateaus is critical. Rehabilitation progress is not linear, and progression should be adapted to match the athlete’s physical response and neurocognitive readiness.22,25
Established rehabilitation strategies such as aquatic therapy (which reduces joint impact while maintaining neuromuscular engagement) and perceptual-neurocognitive training (which targets central reprogramming of movement) offer promising avenues for working through plateaus and enhancing return-to-performance outcomes.8,9,21,26 By presenting two cases the authors aim to:
-
Define and contextualize rehabilitation plateaus within ACL recovery
-
Identify modifiable and non-modifiable risk factors contributing to stagnation
-
Demonstrate the efficacy of adaptive rehabilitation techniques, including gym-based strength, aquatic therapy, and perceptual-neurocognitive programming
-
Advocate for early recognition and intervention to prevent long-term return-to-sport delay or failure
Therefore, the purpose of this case series was to explore clinical rehabilitation plateaus that occur during ACL reconstruction recovery, an underrecognized phenomenon in sports rehabilitation.
Case Description, Subject 1: 36-Year-Old Recreational Athlete Following ACL Reconstruction
Informed consent was obtained from the subject for the use of clinical data and related images.
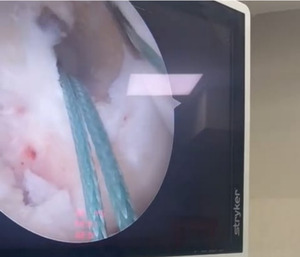
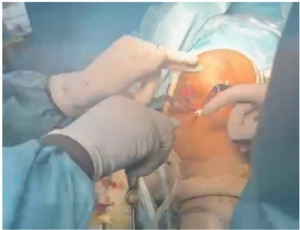
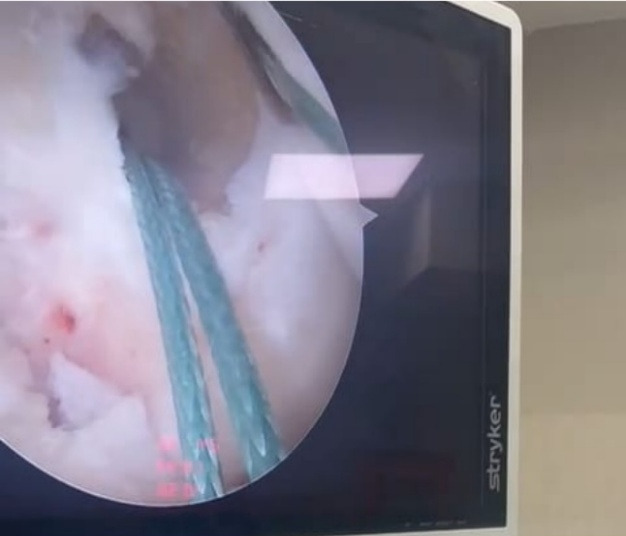
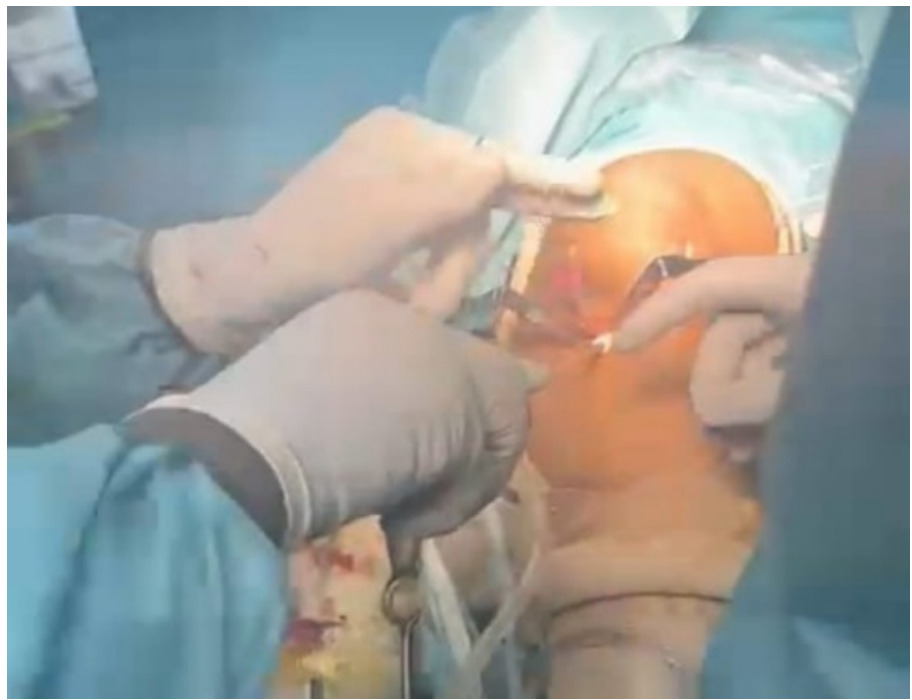
Case-1, a 36-year-old recreational badminton player and private business owner, sustained a non-contact ACL injury on his left knee approximately 10 weeks prior to surgery. The surgical reconstruction utilized a patellar tendon graft, confirmed by intraoperative imaging with the final arthroscopic suture fixation visualized clearly (Figure 1) (Figure 2). The subject was right-leg dominant with no associated injuries. The subject had a prior history of low-level gym training, mostly cardio-focused, and had consistently avoided weight training due to repeated physical discomfort after lifting — including fever, muscle soreness, and nausea-like sensations. This history became a significant barrier during the strength-building phases of rehabilitation.
Following surgery, the subject completed a four-week inpatient and home-based rehabilitation program focused on pain management, early range-of-motion restoration, and quadriceps activation. Structured physiotherapy was initiated approximately four weeks post-surgery, following early therapeutic programming. During the early weeks, his commitment was strong — performing exercises twice daily, including heel slides, SLRs, towel-assisted flexion, patellar mobilization, and wall-assisted movement drills. By the end of Phase 2 of the rehabilitation protocol, the subject had progressed to weight-bearing ambulation with crutches and achieved approximately 100° of knee flexion with full extension.
The subject progressed steadily through Phase 1 (Weeks 1–4) and Phase 2 (Weeks 4–8). These phases included wall-assisted flexion, prone hangs, patellar mobilization, active heel slides, towel-assisted knee flexion, and SLR variations in multiple positions. In the early rehabilitation phase, a modified wooden board-assisted knee flexion exercise was used to re-educate the range of motion (Figure 3). He was able to perform partial weight-bearing gait training and early hip-strengthening drills using resistance bands. The subject achieved full weight-bearing walking with a brace and reached 130° knee flexion by the end of Phase 2.
In Phase 3 (Weeks 8–16), strengthening efforts focused on isolated quadriceps and hamstring work, primarily through isometric quad sets at 0°, 30°, and 60°, resistance band exercises, and supported functional drills such as mini squats, wall squats, and step-ups. The subject also performed cycling, single-leg balance tasks, terminal knee extensions with bands, and calf raises. However, this is where the subject’s fear of weight training became evident. As work demands increased, his exercise frequency dropped from twice daily to once daily and eventually became inconsistent. Despite therapist feedback, he frequently skipped sessions, which limited his progression. His fear of resistance training led to the refusal of free-weight-based exercises.
The subject demonstrated a preference for machine-based resistance exercises and avoided free-weight and dynamically loaded strength tasks, such as barbell squats, deadlifts, and high-load multi-joint drills. His strengthening program therefore remained limited to controlled machine-based exercises, including leg press at 45° and 75° knee flexion angles, seated hamstring curls, and submaximal isometric resistance tasks.
Despite adherence to this modified strengthening approach, objective assessment revealed that he was substantially behind expected strength recovery benchmarks for this phase of rehabilitation. At 7–8 months post-operation, handheld dynamometry demonstrated persistent quadriceps and hamstring strength deficits exceeding 25% compared to the contralateral limb, well below commonly cited return-to-sport thresholds of ≥90% limb symmetry. Functional progression was further limited by reduced tolerance to progressive loading and inability to advance to free-weight or plyometric tasks, indicating delayed neuromuscular and strength restoration.
Phase 4 (Weeks 16–28) introduced more advanced strength and sports-specific tasks. As strengthening progressed, the subject performed resistance band-supported wall squats to improve neuromuscular control and weight acceptance (Figure 4), bilateral and single-leg squats at various angles (30°, 60°, 90°), split squats, lateral band walks, lunges, mountain climbers, balance training on unstable surfaces, and step-ups with variable box heights. He also attempted speed-walking and light jogging drills. During this period, the subject remained consistent with exercises like light dumbbell squats, single-leg stance balance work, ball squeeze drills for VMO activation, and agility tasks on level surfaces. However, The subject avoided performance of resisted lunges, power-based single-leg drills, and explosive tasks due to discomfort and psychological hesitation. Although he engaged in the program, his overall intensity and load progression were limited.
Between Weeks 24 and 28 post-operation, progression in strength and functional capacity began to slow; however, this was initially obscured by the subject’s continued tolerance of low-load, machine-based strengthening exercises. By approximately Week 31 (7 months post-operation), the plateau became clinically evident when repeated reassessments failed to demonstrate further improvements in quadriceps and hamstring strength despite ongoing rehabilitation participation.
The subject also reported a qualitative change in post-training response. Earlier in rehabilitation, post-exercise muscle tightness was perceived as indicative of effective muscle activation and adaptation. During this later phase, however, similar sensations were transient and no longer translated into sustained strength gains or functional improvements. The combination of persistent strength asymmetry, limited progression beyond machine-based loading, and lack of measurable improvement despite continued rehabilitation led to formal identification of a rehabilitation plateau—defined as stagnation in physical and neuromuscular progress despite adherence to a structured protocol.
Throughout all phases of rehabilitation, aquatic therapy was incorporated as a complementary method. In the initial phase, aquatic-based movement helped the subject regain strength and coordination with reduced joint load. Underwater walking, supported squats, and leg movements allowed pain-free activation of key muscle groups. As rehabilitation advanced, hydrotherapy sessions were used for active recovery post-strength training and to support cardiovascular reconditioning. These sessions improved overall comfort and allowed the subject to train without fear of injury.
These previously identified factors avoidance of higher-load resistance training and inconsistent rehabilitation attendance persisted during this phase and contributed to the sustained lack of strength adaptation. He was mentally fatigued, frustrated by the lack of improvement, and became hesitant to continue sports-specific drills. Despite completing the full structured protocol, his progression from partial strength to full strength never fully occurred. This transition, often the most demanding and decisive part of post-ACL rehab, became the stage where the rehabilitation plateau became fully apparent.
Clinical Decision-Making and Program Modification Following Plateau Identification
Upon identification of a rehabilitation plateau, the treating therapist modified the rehabilitation program to better align with the subject’s physical tolerance, psychological readiness, and observed neuromuscular deficits. Given the subject’s continued avoidance of free-weight exercises, the program emphasized tolerated machine-based resistance training (e.g., leg press and hamstring curls), prolonged isometric holds at 30°–90° of knee flexion, and lateral walking drills using resistance bands. Heel raise exercises with resistance bands were incorporated to improve calf activation and lower-limb coordination during late-stage rehabilitation.
To address persistent proprioceptive and coordination deficits, additional interventions included ball-control tasks, balance board exercises, and step-down drills. Modified agility activities—such as controlled lateral stepping, stair training, and sustained squatting on uneven surfaces—were introduced to safely re-expose the subject to functional loading demands. Aquatic therapy was continued as an adjunct for active recovery, circulation enhancement, and pain-free movement exposure. Collectively, these targeted modifications were intended to maintain functional capacity, address neuromuscular limitations, and prevent further regression while respecting the subject’s load tolerance and engagement level.
Outcomes
At the 12-month review, the subject had no complaints of pain (VAS: 0). Thigh girth was measured using a standard flexible measuring tape at a fixed distance proximal to the superior pole of the patella, with the subject positioned supine and the limb relaxed. Quadriceps girth increased from 49.0 cm to 51.2 cm over the course of rehabilitation. While girth measurement does not directly quantify muscle strength or neuromuscular control, it was used in this case as a supplementary indicator of muscle mass restoration.
Functional strength and motor control were primarily inferred through single-leg squat quality, step-down performance, balance task execution, and tolerance to progressive loading during functional drills, rather than girth alone. Persistent deficits in single-leg control, endurance during closed-chain tasks, and inability to progress to higher-load or dynamic activities were key clinical indicators used to identify the rehabilitation plateau. The subject was able to walk independently, climb stairs, and carry out daily tasks, however, he could not jog or return to sport comfortably. The subject made a personal decision to step away from competitive badminton and instead engage in the sport recreationally for fitness and enjoyment.
This case illustrates a clinically observable ACL rehabilitation plateau, characterized by stagnation in strength progression, loading tolerance, and functional advancement despite completion of a phase-wise rehabilitation program. Between postoperative months 6 and 8, the subject demonstrated minimal improvement in functional task tolerance and was unable to progress beyond partial strength gains. Although quadriceps girth showed only marginal change during this period, functional performance deficits and limited tolerance to increased loading were the primary indicators used to identify the plateau.
The plateau appeared multifactorial in nature, influenced by limited exposure to high-resistance training, psychological apprehension toward load progression, and inconsistent rehabilitation attendance related to occupational demands. Motivation was not quantified using a formal psychometric scale; however, reduced engagement was clinically inferred from inconsistent participation, reduced exercise adherence, and expressed frustration during advanced rehabilitation stages. Identification of the plateau allowed for targeted program modification, which helped preserve functional capacity, although full return-to-sport readiness was not achieved within the study period.
Case Description, Subject 2: 16-Year-Old Sub-Elite Football (soccer) Player Following ACL Reconstruction
Informed consent was obtained from the subject for the use of clinical data and related images. The subject of case 2 was16-year-old sub-elite football (soccer) player, who sustained a contact ACL injury on his left knee during a defensive tackle in a competitive match. The subject was immediately removed from the field due to knee swelling, pain, and instability. MRI confirmed a complete ACL tear with no additional ligament or meniscal damage. He underwent ACL reconstruction using a patellar tendon graft. Prior to injury, the subject regularly participated in district- and school-level tournaments as a central midfielder and was highly committed to his football career.
Post-surgery, the subject’s rehabilitation was initially managed by the physiotherapy team associated with his football club. While the early phase included standard components such as brace-supported walking, ankle pumps, assisted heel slides, static quadriceps activation, TheraBand drills, cycling, and proprioceptive balance exercises, the program lacked progression, load management, and structured neuromuscular re-education. Although well-intended, the initial rehabilitation did not adequately match the performance demands of a young sub-elite athlete. After three months of basic rehabilitation, the club sessions were concluded, and unfortunately, the athlete did not receive further structured rehabilitation for the following three months. Importantly, despite resumption of structured rehabilitation after this interruption, the athlete demonstrated persistent deficits in neuromuscular timing, perceptual processing, and sport-specific movement efficiency, indicating a true rehabilitation plateau rather than a simple delay in care.
This three-month gap occurred during a critical strength and neuromuscular development window, resulting in a significant delay in physical progress and return-to-sport readiness. When the subject presented to the authors’ physiotherapy facility approximately seven months postoperatively, he was pain-free but displayed clear signs of physical deconditioning.
The clinical examination revealed visible quadriceps atrophy, poor balance, reduced single-leg control, diminished muscular strength, asymmetry during squatting tasks, delayed muscle response during dynamic drills, and hesitation while attempting directional changes. Explosive mechanics tasks requiring rapid force production and neuromuscular coordination required for dynamic tasks such as jumping, sprint initiation, deceleration, and change-of-direction movements were diminished. The subject reported that he did not feel “stable” or “strong” when attempting to perform high-speed football movements. Muscle strength was assessed isometrically using a handheld dynamometer. At eight months post-surgery, the injured limb quadriceps strength measured 16.8 kg and hamstring strength 18.5 kg, compared to the uninvolved side quadriceps 22.4 kg and hamstring 21.3 kg. (Table 1)
Despite following a standard rehabilitation plan earlier, it was clinically observed that the subject demonstrated persistent deficits in movement quality, single-limb control, and confidence during sport-specific tasks, suggesting reduced readiness required for return to sport. Following re-entry into structured rehabilitation, deficits persisted in neuromuscular control and explosive function, as evidenced by poor single-leg stability during dynamic tasks, delayed force generation during push-off, and impaired coordination during change-of-direction activities. Functional assessment revealed reduced single-leg control during hop and landing tasks, diminished reactive response during externally cued agility drills, and inconsistent movement sequencing during sport-specific activities.
Although training exposure and attendance improved after resumption of care, the athlete failed to demonstrate expected progression in explosive lower-limb function or neuromuscular integration required for high-speed football actions. These findings indicated that the rehabilitation plateau reflected a prolonged stagnation in neuromuscular and motor control recovery rather than insufficient rehabilitation duration alone. The plateau was therefore characterized primarily by impaired brain–body integration, limiting effective force production, reactive control, and sport-specific movement execution.
Clinical Decision-Making and Program Modification Following Plateau Identification
A new rehabilitation strategy was initiated when this subject began therapy with the authors, focusing first on regaining isolated strength and neuromuscular control. Strength exercises included machine-based leg press, hamstring curls (progressed from isometric to eccentric), Spanish squats, step-ups with increasing resistance and height, Romanian deadlifts, single-leg wall sits, and balance board drills.Upon resumption of therapy, the patient progressed through foundational motor tasks, including jogging in place (Figure 5), to reintroduce neuromotor demand under controlled conditions. Given the patient’s age, recovery potential, and demonstrated engagement with rehabilitation, gradual progression to gym-based resistance training was initiated under close supervision.
Strength progression was monitored through repeated clinical strength testing and functional task performance. Following introduction of controlled barbell squats, deadlifts, and dumbbell-based single-leg exercises, reassessment demonstrated improved symmetry during bilateral and unilateral loading tasks, increased tolerance to external resistance, and improved movement quality during squatting and step-down activities. Additionally, single-leg functional tasks showed improved control and reduced compensatory strategies compared with earlier assessments, supporting observed improvements in quadriceps activation and overall lower-limb strength.
Recognizing that the athlete’s performance was limited by more than strength, the program was further advanced with perceptual-neurocognitive training. These drills aimed to rewire brain-body integration, sharpen field awareness, and retrain reactive movement patterns. Dual-task drills incorporating visual-cognitive load, such as ball reception with cone awareness, were introduced to simulate sport-specific coordination (Figures 6, 7).
Aquatic therapy was used throughout the program to assist with joint unloading and active recovery. Water-based walking, squatting, and light leg movements in the pool allowed him to train safely on off days, reducing joint fatigue and supporting psychological confidence.
This foundational phase after time away from therapy served to restore baseline strength and movement coordination, but more advanced and individualized strategies were needed to address the plateau and reintroduce sport-specific performance.
The subject progressed to performance of wall-based laser pointer tracking to address control of knee alignment, externally cued foot-tap drills requiring rapid visual stimulus recognition and immediate lower-extremity response, and dual-task exercises that combined agility with verbal or memory cues. These strategies helped challenge his mind and movement simultaneously, preparing him for the dynamic, high-stimulus nature of football.
In the later stage of rehabilitation, specific focus was given to position-specific perceptual drills, tailored for his role as a central midfielder. Verbal-cue-based lateral drills, tailored to his midfield football role, were crucial for restoring functional agility (Figure 8). 360° cone scanning with ball reception, directional passing under time constraints, agility-based decision-making tasks, and reactive running with changing cues. Controlled ball handling under visual distraction and small-space turn-and-pass combinations were also integrated. These drills improved his sport perception, helped restore his timing and vision, and gradually reconnected him to real-match demands.
Outcomes
By the end of the ninth postoperative month, the athlete demonstrated measurable improvements in lower-limb strength and functional performance. Reassessment revealed improved symmetry during loaded squatting tasks, enhanced control during single-leg hop activities, and improved tolerance to repeated agility-based movements with reduced fatigue. These functional gains coincided with improved confidence during change-of-direction tasks and increased engagement in the sport-readiness phase of rehabilitation.
Objective strength testing using a handheld dynamometer demonstrated progressive improvement in quadriceps and hamstring strength over time, with restoration of limb symmetry by 12 months post-surgery (Table 1).
By the 14th to 15th month, the subject successfully re-entered team training under supervision, first in non-contact sessions and then in full-contact play. At the time of final review, he had returned to competitive football with no pain, good quadriceps symmetry, strong perceptual control, and renewed field confidence. The subject continued periodic neurocognitive and perceptual drills as part of his long-term performance maintenance plan.
This case represents a clear example of a rehabilitation plateau in a youth athlete, triggered not by reinjury or surgical complications, but by a lack of progressive overload, insufficient neuromuscular challenge, psychological withdrawal, and a critical gap in training continuity. The athlete’s initial recovery stalled not because of lack of effort, but due to an early-stage program that lacked specificity and later disruption in rehabilitation.
The use of advanced techniques such as perceptual-neurocognitive drills, position-specific movement training, and active recovery through aquatic therapy helped re-engage his brain-body connection and athletic confidence. This multifactorial, individualized approach proved essential in overcoming the plateau and guiding a successful return to sport. The plateau was only overcome after the introduction of targeted perceptual–neurocognitive training and position-specific reactive drills, further supporting that the stagnation reflected a central neuromuscular limitation rather than the earlier interruption in care alone. To address the identified plateau, rehabilitation focus was shifted from volume-based strengthening to targeted late-stage strategies emphasizing neuromuscular timing, perceptual processing, and task-specific loading. This approach aimed to reintroduce adaptive stimuli capable of overcoming the observed stagnation.
DISCUSSION
Rehabilitation following anterior cruciate ligament (ACL) reconstruction is often conceptualized as a linear progression through predefined phases; however, the present cases illustrate that recovery trajectories are frequently non-linear. Both cases demonstrate that rehabilitation plateaus may emerge when the applied rehabilitation stimulus fails to match the evolving biological, neuromuscular, or psychological needs of the athlete. Rather than reflecting failure of surgery or non-compliance, plateaus in these cases represented periods of insufficient adaptive stimulus relative to recovery stage.
In Case 1, the rehabilitation plateau appeared to develop gradually during the transition from isolated strengthening to higher-load functional demands. Although early rehabilitation progressed appropriately, the patient’s long-standing aversion to resistance training limited exposure to progressive overload. Psychological apprehension regarding weight training, compounded by occupational stress and inconsistent training frequency, likely contributed to subthreshold mechanical loading. From a physiological standpoint, insufficient load variability and intensity may have limited neuromuscular adaptation, particularly in late-phase strength and power development. Earlier identification of stagnation—through systematic monitoring of strength progression, load tolerance, and functional transfer—may have allowed targeted education addressing fear-avoidance beliefs and gradual exposure to resistance training before strength deficits became entrenched.
In contrast, the plateau observed in Case 2 was abrupt and temporally linked to a prolonged interruption in rehabilitation during a critical neuromuscular development window. The absence of structured loading and sport-specific stimulation during early recovery likely impaired motor pattern consolidation and neurosensory integration. When rehabilitation resumed, deficits were evident not only in strength but also in reactive control, movement sequencing, and perceptual-motor coordination. This suggests that the plateau in Case 2 was driven less by psychological resistance and more by delayed neuromuscular maturation resulting from early under-stimulation. Importantly, youth status alone did not confer protection against stagnation when rehabilitation continuity and task specificity were insufficient.
The differing mechanisms underlying plateau development in these cases underscore that rehabilitation stagnation is not a uniform phenomenon. In Case 1, the plateau reflected a failure to progress from partial to high-load strength adaptation, whereas in Case 2, it reflected impaired translation of neuromuscular capacity into sport-specific performance. Recognition of these distinctions is critical, as intervention strategies must be aligned with the dominant limiting factor—whether mechanical loading tolerance, neuromuscular integration, or perceptual readiness.
Intervention strategies were therefore tailored to the underlying contributors in each case. In Case 1, program modification focused on restoring confidence with loading through controlled resistance exposure, isometric holds, and proprioceptive exercises, supported by aquatic therapy to reduce joint stress and improve tolerance. Despite functional improvement, the absence of sustained high-load exposure limited progression to full sport readiness. In Case 2, rehabilitation emphasized restoration of neuromotor timing and perceptual-motor integration through dual-task training, externally cued agility, and position-specific decision-making drills. This approach successfully addressed the neurocognitive contributors to stagnation and facilitated return to competitive sport. A comparative overview of patient characteristics, rehabilitation trajectories, plateau mechanisms, and post-plateau interventions for both cases is provided in Table 2.
Collectively, these cases highlight that rehabilitation plateaus should be viewed as clinical signals indicating a mismatch between rehabilitation stimulus and athlete readiness rather than as endpoints of recovery. Earlier identification—through systematic assessment of strength progression, functional transfer, and neuromuscular control—may allow clinicians to intervene proactively. Addressing plateaus requires flexibility in program design, targeted education, and integration of strength, neurocognitive, and perceptual-motor strategies. Recognizing and responding to stagnation may help optimize recovery trajectories and reduce prolonged or incomplete return-to-sport outcomes. From a clinical perspective, recognizing rehabilitation plateaus as checkpoints rather than failures allows clinicians to reassess loading strategies, neuromotor demands, and psychological readiness, and to adapt rehabilitation accordingly rather than relying on rigid, time-based progression. The key contributing factors associated with rehabilitation plateau development in both cases are summarized in Table 3.
This case series is limited by the utilization of only two cases, and the absence of validated patient-reported outcome measures to formally assess outcomes and psychological or motivational factors contributing to rehabilitation plateau. Additionally, follow-up was limited to the short-to-medium term, and long-term outcomes such as sustained sport participation or re-injury risk were not evaluated. Future investigations should explore rehabilitation plateaus in larger cohorts, incorporate objective strength and neuromuscular assessments alongside psychological screening tools, and evaluate targeted interventions aimed at identifying and addressing stagnation earlier in the rehabilitation process.
CONCLUSION
Rehabilitation following anterior cruciate ligament reconstruction is not always linear, and structured, time-based protocols alone may fail to accommodate individual variability in physical, neuromuscular, and psychological adaptation. The two presented cases illustrate that rehabilitation plateaus can emerge despite protocol adherence, reflecting a mismatch between rehabilitation stimulus and athlete readiness rather than surgical failure or non-compliance. These plateaus manifested through distinct mechanisms in each case, including resistance to progressive loading, disrupted rehabilitation continuity, and delayed neuromuscular or perceptual-motor integration. Early recognition of stagnation through systematic reassessment allows clinicians to modify rehabilitation strategies by adjusting loading parameters, incorporating neuromotor and perceptual demands, and addressing psychological barriers. Viewing the rehabilitation plateau as a clinical inflection point rather than a setback may facilitate more individualized, outcome-driven rehabilitation and improve return-to-sport readiness following ACL reconstruction.
CORRESPONDING AUTHOR
Ram Kumar E., MPT (Sports Physiotherapy)
Sports Physiotherapist and Clinical Researcher
Chennai, Tamil Nadu, India
Email: kumra292000@gmail.com
ORCID: [https://orcid.org/0009-0002-2357-2389]
Acknowledgment
The authors thank the patients for their participation and consent, and acknowledge the contributions of co-therapists, mentors, and peers who provided clinical support and constructive feedback during the development of this manuscript.
Conflict of interest
The authors declare no conflict of interest.
Funding
No external funding was received