INTRODUCTION
Partnerships between clinicians and researchers are vital to ensure that research addresses practical and clinically relevant issues in patient care. These partnerships also promote the translation of scientific discoveries into practice, allowing new treatments, technologies, and methodologies to be rapidly adopted in clinical settings.1 Given the importance of clinician-driven scholarship and the experience of our research group in developing thriving partnerships between clinicians and researchers, this commentary aims to provide a roadmap for successful clinician-driven research through the lens of sports medicine research. The respective institutions of the authors include: Two pediatric hospitals with affiliated university medical training, a large public university with an affiliated academic medical center, and a private university located in different regions of the United States. Drawing on the experiences and lessons learned from collaborative efforts, the authors will explore:
-
Common barriers preventing clinicians from engaging in research, such as time constraints, lack of training, and insufficient institutional support.
-
Strategies to overcome barriers, including structured mentorship, building collaborative networks, and embedding research into routine clinical practice.
-
A roadmap for successful clinician-driven research, highlighting the key components needed to foster active collaboration, generate impactful scholarship, and ensure continuous quality improvement.
The purpose of this commentary is to highlight the barriers clinicians face when engaging in research and proposes practical strategies to foster collaborative partnerships between clinicians and researchers.
Defining Success in Partnerships between Clinicians and Researchers
Success in partnerships between clinicians and researchers can be measured through impactful scholarship, active multidisciplinary collaborations, continuous quality improvement or assurance, and the development of clinician-scientists within your organization. Key successes include the development of in-clinic assessment programs at the authors respective institutions that allow clinicians to gather data with dual purposes—supporting clinical decisions about return to play after injury and contributing to rigorous clinical research. In most cases, these programs bring together expertise from clinicians and researchers to include both feasible and evidence-based physical and patient-reported outcomes that are integrated directly into the flow of patient care. This approach ensures that data generated is not only beneficial for immediate patient care by clinicians but also forms a basis for peer-reviewed publications and presentations at national conferences in the hopes of positively influencing patient care outside of our local contexts. As a result, the research teams have observed meaningful changes in clinical care (e.g., longer times to clearance for return to play after anterior cruciate ligament reconstruction) and consistently published results through collaborative, multidisciplinary teams, uniting experts from orthopedics, biomechanics, physical therapy, and rehabilitation. These partnerships have allowed investigation into complex questions in ACL injury recovery and rehabilitation with a holistic perspective, ultimately leading to more effective patient outcomes and enhanced research impact. Perhaps most significantly, these partnerships have led to the creation of the ACL Reconstruction Rehabilitation Outcomes Network (ARROW). ARROW is a multi-site research registry focused on fostering clinician-scientist development while producing high-impact, clinically relevant research (https://acldashboard.com). By pooling resources and data across institutions, this model allows for research that is both scalable and impactful across diverse clinical settings.
BARRIERS AND SOLUTIONS TO ENGAGING IN CLINICAL RESEARCH
While clinician-scientists offer tremendous value in the promotion and practice of evidence-based medicine, they have been described as an “endangered species,” due to the decrease in the number of clinicians who engage in research.2 Several works have explored the barriers to a clinician’s engagement in research which appear to differ based on the healthcare profession.3–5 Specifically, in the field of sports medicine, Sugimoto et al. reported different barriers to research engagement between sports medicine physicians and allied health practitioners.3 While lack of time was reported as the top barrier to research engagement for both physicians and allied health practitioners, allied health practitioners reported unique barriers including feeling the lack of authority to implement research findings into clinical practice, lack of perceived benefit to their own professional career, and limited funding to support research activities.3 Furthermore, allied health practitioners reported the availability of a research mentor as the greatest facilitator to their engagement in research.3 While it is important to note that the work of Sugimoto et al. was limited to the perceptions of a single sports medicine society, these findings may have a broader application. McCarty et al. reported similar findings in a study of barriers toward evidenced based practices among athletic trainers,6 as well as inadequate time as the most frequently reported barriers.6 The identification of these barriers prompts the need for tailored solutions to promote increased engagement and ultimately revive the role of the clinician-scientist. Factors that influence research engagement can be categorized as clinician based, social and cultural, and institutional.
Hurdles to Research Engagement Experienced by Clinicians
Many rehabilitation providers are exposed to research through article reviews, methodology courses, and limited participation in studies during education and clinical training. However, despite interest in research, many clinicians may feel unprepared to lead research endeavors likely due to a lack of knowledge and training, leaving them overwhelmed about where to start. The perceived value of research engagement may vary by clinical setting and leadership priorities, which may lead to a focus on professional development that directly enhances clinical skills and patient visits. Additionally, declining reimbursement7,8 and the demands of clinical documentation, especially in electronic medical record (EMR) systems, make it difficult for clinicians to balance research with their productivity requirements.
Overcoming Clinician-level Hurdles to Research Engagement
Despite these barriers, there are numerous strategies clinicians can employ to successfully engage in research. By leveraging mentorship, utilizing local and online resources, and carefully preparing for data collection, clinicians can take meaningful steps towards contributing to evidence-based practice (EBP).
New Research Efforts are Challenging and Will Not Always be Linear
It’s important to acknowledge that the research process is often nonlinear, requiring patience and resilience to navigate the challenges that arise. Most allied health providers lack protected research time as part of their job description. The clinician-scientist should discuss with their leadership ways to maximize clinical productivity while carving out dedicated time to allow for research projects. The beginning clinician-scientist may need to make some tradeoffs in the early stages, such as using lower volume times in clinic to work on research projects. Finally, as clinical questions develop and are answered, it is imperative to find time to present these findings to help report new discoveries, develop new questions, and ultimately change clinical practice.
Partnering with a Mentor or Experienced Researcher
It is the belief of the authors that partnering with a mentor or experienced researcher is one of the most effective strategies to overcome the barriers clinicians face when engaging in research.9 To identify potential mentors, clinicians can begin by exploring local academic institutions, research centers, or professional organizations in their field. Many experienced researchers are listed as faculty members on university websites or as authors on published research in professional journals, making them easy to identify. Professional conferences also provide opportunities to network with researchers who share similar interests. When contacting a potential mentor, clinicians should be specific about their research interests, current clinical questions, and goals for the collaboration. Importantly, many experienced researchers often struggle to find clinicians who can provide the clinical expertise and access to patient populations that are crucial for applied research. Reaching out to a researcher with a clear proposal for collaboration can be well-received and mutually beneficial. Researchers gain the opportunity to work with real-world clinical data, while clinicians benefit from the guidance and structure needed to initiate and conduct meaningful research.5
Local and Online Resources
Prior to the clinician or clinician-scientist initiating a clinical research project, it is imperative to understand what education, technology and resources are available to help support the process. Some examples that may be available to you at no or very low cost include:
-
Clinical and Translational Science Institutes (CTSIs): Many academic medical centers have CTSIs that provide resources like grant writing workshops, research design consultation, and biostatistical support, often free or at a reduced cost for clinician-researchers.
-
Professional Society Research Networks: Many professional organizations (e.g., American Physical Therapy Association, National Athletic Trainers Association) have formal research networks or research groups that offer mentorship programs, online databases for literature searches, and access to data repositories for secondary data analysis.
-
Institutional Review Board (IRB) Training and Mentorship: Many institutions IRBs offer training programs and mentorship to help clinicians navigate the process of initiating research projects and maintaining compliance with best practices. These resources can be instrumental in ensuring ethical standards and regulatory compliance.
-
Online Educational Platforms: Websites like Coursera, edX, and NIH RePORTER offer courses and tools in research methodology, data analysis, and scientific writing. These can be invaluable for clinicians who need to refresh or expand their knowledge on conducting research.
-
EMR Integration Tools: Tools like REDCap or specific EMR features (e.g., data extraction capabilities) can help streamline data collection and analysis directly from clinical settings, reducing the burden on clinicians.10,11
-
Open Access Journals and Preprint Archives: Resources like the Journal of Athletic Training, PubMed Central and medRxiv provide easy access to current literature, facilitating clinicians’ engagement with the latest research and allowing for dissemination of their own findings in open-access formats.
These additional resources can offer practical ways to overcome logistical and educational hurdles, enabling clinicians to successfully engage in meaningful research projects.
Setting Yourself Up for Successful Data Collection
Data collection is a critical but often overlooked area for the clinician-scientist working in a busy clinical setting. For this reason, clinical tools and technology can help reduce errors and maintain security compliance in data collection. The popularity of REDcap, a secure platform for managing online research databases and surveys, has allowed for easier data collection.10,11 Using data validation, a method of providing constraints on data entered in individual fields, in REDcap and other data collection tools like Microsoft excel will assist in data accuracy and save time. Patient reported outcome questionnaires, often a mainstay of clinical research, can be captured using QR code scanning on smartphones or in-house tablets. Many EMR systems, including EPIC, allow for customization of documentation fields, as well as syncing the EMR with a secure online database to reduce redundant data entry (i.e., Epic Clinical Data Pull). For example, a clinician-scientist may be able to develop standardized charts for strength assessments to allow all clinical providers to enter data in as part of their daily documentation workflow which can also be used for research purposes. At larger institutions, data engineers can be of value for larger data queries. Many allied health clinics will have support staff that are working to gain clinical experience in a gap year prior to beginning graduate studies. Engaging these staff members in assisting with data entry, abstract development, data visualization and even patient consenting can assist your projects as they build their resume and gain valuable experience.
Social and Cultural Hurdles to Research Engagement
Creating a culture of clinical inquiry is described as the first step in the EBP model.12 The culture of the work environment in which a clinician practices will likely have a strong influence on their ability to engage in research. A culture that does not support and reward evidenced based care will make EBP unsustainable.13,14 Further, workplace culture that places a disproportionate priority on patient volume and clinical productivity may negatively impact the practice of evidence-based medicine as well as the priority placed on clinical scholarship. With time consistently reported as a barrier to research engagement and EBP, creating a culture that places equal value on volume/productivity and quality of care/evidence-based medicine is necessary before research partnerships can thrive. Furthermore, the field of sports medicine can often create a culture of “authority” where practitioners are trained to think less creatively and with less curiosity and rely more on their superiors for direction.3 It is the opinion of the authors that this culture may dissuade practitioners from questioning the current standard of practice and thus serve as a roadblock to the generation of good clinical questions and subsequent research engagement.
Proposed Strategies to Overcome Social and Cultural Hurdles
Behavior modeling has widely been accepted as a successful method to illicit behavior change.15 Thus, creating a culture of clinical inquiry should start with the modeling of this behavior from institutional leadership (e.g., service line directors, senior clinicians). Leaders must set an example and demonstrate the behaviors they wish to see in their staff, this will in turn, set the standard and influence staff to adopt the same behaviors.5 The authors have successfully promoted a culture of inquiry by organizing team retreats focused on learning and collaboration. Senior clinicians are responsible for planning the retreats, which center on topics relevant to patient care. They present new evidence and lead group discussions, while also training less experienced clinicians in evidence-based interventions that may be new to them. Novice clinicians then apply this knowledge in their own practice, helping to ensure that the latest evidence-based approaches are integrated into patient care.
Previous work and academic models have identified the need for administrative support and time to allow the development and implementation of evidence-based practice.16,17 Administrative healthcare leaders can demonstrate their value of evidenced-based medicine by providing clinicians with adequate time to engage in EBP during working hours. This may involve hosting regular journal clubs during the work week that encourage all staff to attend and present. This provides clinicians with an opportunity to review and discuss new evidence as a team, which may naturally lead to a conversation amongst staff and leaders on how to best implement the evidence into clinical practice. This may also spark additional questions related to the journal club topic that might be pursued through point of care research. Leaders should also consider setting aside a few hours each week for clinicians to participate in EBP projects. Using naturally slower periods during the day can benefit both the clinician and the organization. These projects might include collaborating with physicians to develop evidence-based rehabilitation protocols for the institution, conducting chart reviews to evaluate the quality of current clinical practices and the application of evidence-based interventions, or engaging in research projects.
Lastly, leadership can show their support of research engagement by encouraging clinicians to attend and present at local and national conferences. This can be in the form of providing financial support for registration and travel costs, providing paid time off for attendance, or providing incentives such as bonus or promotion opportunities that favor research engagement. One way this group has successfully promoted clinician engagement in EBP is through the implementation of a clinical ladder. A clinical ladder is a structured framework designed for professional development, job satisfaction, and retention.18 Originating in the field of nursing, a clinical ladder requires practitioners to complete tasks across multiple domains including clinical, educational, leadership, and research. The program offers incentives, including promotion, for job performance beyond the basic requirements. The clinical ladder has been described in nursing literature as an effective method for enhancing nurse competencies, EBP involvement, and retention.19 By designing a clinical ladder that prioritizes participation in EBP initiatives and research, clinicians are encouraged to engage in these activities, fostering a culture that values EBP.19
Institutional Hurdles to Research Engagement
One of the most consistently reported institutional barriers to research engagement is the lack of time.3,20 It has been the experience of the authors that clinicians face pressure to meet volume-based productivity standards, which are often tied to their performance evaluations, leaving little room for research activities. The fast-paced clinical environment further limits opportunities for research. Additionally, insufficient research funding is a common obstacle, either due to direct costs like IRB fees, recruitment, or data collection, or indirect costs such as equipment, space, and loss of revenue from non-billable research time. Without proper funding, clinician-driven research becomes difficult. Moreover, some institutions lack research infrastructure, and those that do may not effectively educate clinicians about available resources or processes for conducting research. Customized solutions are necessary for research to thrive in these environments.
Proposed Strategies to Overcome Institutional Hurdles
Clinician-scientists should be mindful of their institution’s financial limitations and start with low-cost research initiatives. Open communication with leadership about research ideas and progress is essential, along with finding creative ways to allocate time for research, such as scheduling blocked administrative time during non-peak hours. Retrospective chart reviews, survey studies, and case reports are ideal starting points when funding is limited. Leadership can assist by securing alternative funding sources, such as donor funds through institutional foundations, to support research without negatively impacting the department’s financial health. These efforts can help clinicians establish a solid foundation for future research projects.17
For institutions with robust research infrastructure, clinicians should take advantage of educational opportunities to understand available resources and processes for initiating studies. Organizations that actively support clinician involvement in research can provide time or resources, enabling more staff to participate. In settings without this infrastructure, leadership should encourage collaborations with other institutions that have the necessary resources. Multi-center studies, research mentorship, or contributing to existing studies in an indirect role are viable options. Clinicians can also build networks by participating in professional organizations, which can lead to collaborations with researchers who have more experience or institutional support. These collaborations may include IRB reliance agreements and joint study designs, where one institution handles data collection and analysis, easing the burden on others. By working together, clinicians without direct research infrastructure can contribute meaningfully to evidence-based practice while fostering their own research development.
KNOWLEDGE TO ACTION – GETTING STARTED WITH RESEARCH COLLABORATION
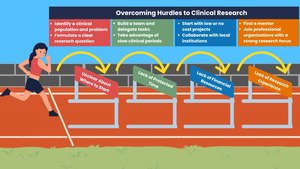
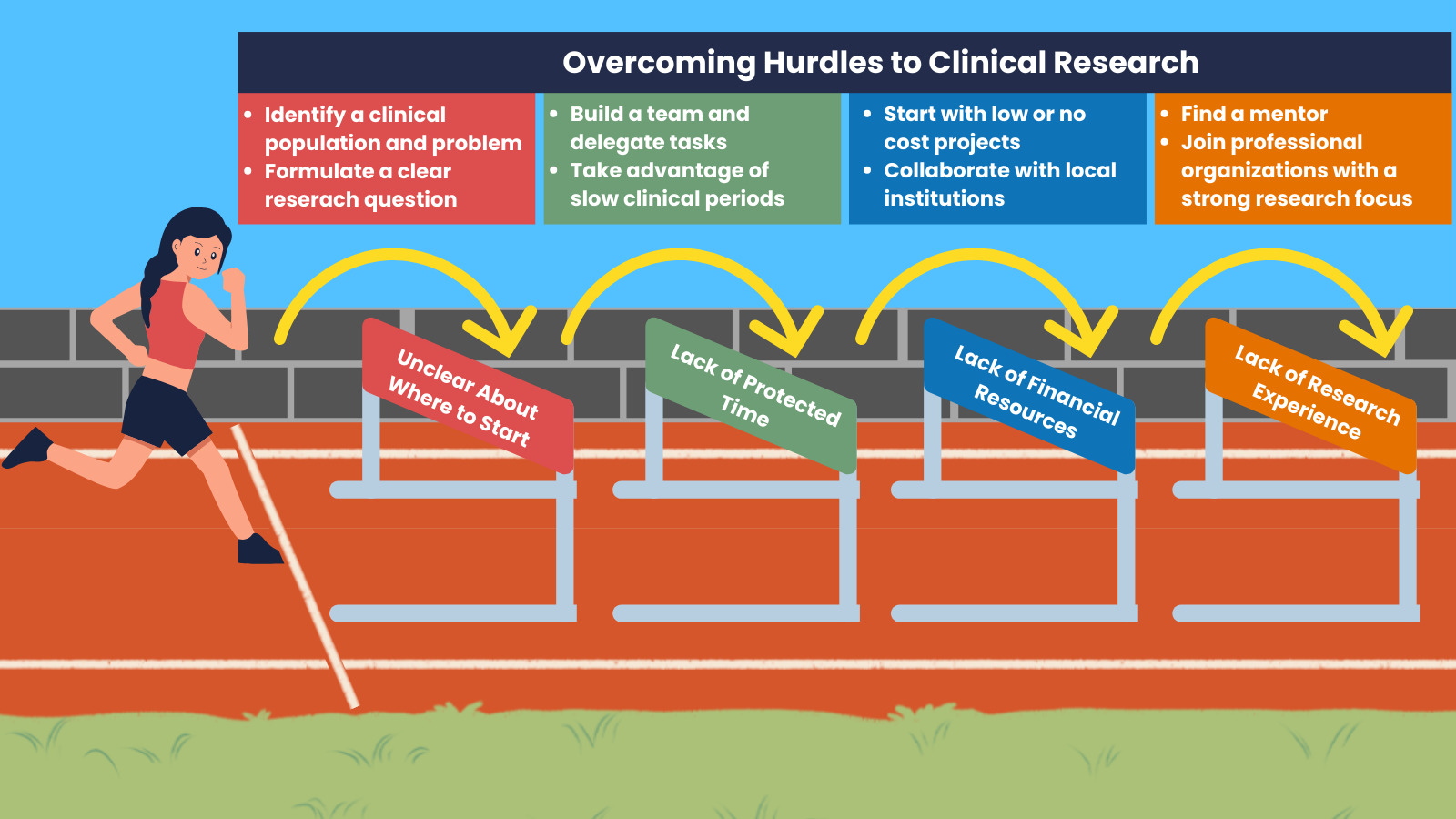
One of the most challenging aspects of engaging in research as a clinician is determining how and where to begin. This decision can feel overwhelming, highlighting the need for a clear framework for success. Drawing on the combined experiences of clinician-led researchers, a roadmap has been developed to assist allied health practitioners in navigating the research process as novice clinician-scientists (Figure 1).
- Identifying a clinical problem and formulating a clear research question - As a novice clinician-scientist, it is easy to create a question that is overly broad and complex, making it difficult to address through clinical research. If the data collection process is too extensive or requires significant effort beyond routine tasks, it will hinder the likelihood of success. To avoid this pitfall, practitioners should begin with smaller projects that focus on one or two specific outcome variables that can be easily collected within their daily workflow in an easily accessible patient population. Being overly ambitious and jumping into a large-scale study, such as a clinical trial, is likely to lead to failure.
-
Determine the feasibility of your research question - Practitioners must assess the feasibility of answering their research question based on the resources available at their institution. All research incurs costs and requires dedicated time. Before beginning a project, it is essential to ensure that the leadership is informed and agrees to cover these costs. When approaching conversations with administrators, our team has found it especially helpful to focus the potential for research to improve patient care and the professional development opportunities that may result from an active research agenda.
-
Assembling a research team - As a busy clinician, conducting a study independently may not be practical. When forming a team, you will need members with strong research design skills to guide the project, someone with statistical expertise for data analysis, individuals for data collection and recording, and those experienced in manuscript writing. If individuals with these skills are not available at your institution, professional organizations and local universities or research medical centers can be a great resource when team building. Building a well-rounded team with clear roles will help reduce the clinician’s workload and increase the chances of success.
-
Identify a mentor and develop a relationship - Mentors come in many forms, so it is important to find one that suits your specific needs. It’s advisable to recognize your own weaknesses in the research process and seek a mentor who can address those gaps. Clinicians shouldn’t feel restricted to choosing a mentor from their own institution and should remain open to seeking mentorship from external sources. Engagement in professional organizations with a research focus is a great way to connect with potential research mentors.
-
Establish realistic timelines - Research is a lengthy process, often taking one to two years (or more) for a study to progress from inception to completion and publication. Unrealistic expectations for the pace of research are among the most common pitfalls the authors have experienced when working with novice clinicians and scholars who are attempting to engage in multidisciplinary research. Understanding the steps involved and the typical timeframe for each phase is crucial to prevent frustration and discouragement when progress feels slow.
CONCLUSION
Fostering clinician-researcher partnerships is essential to advancing evidence-based practices and improving patient care. While clinicians face several barriers to engaging in research, strategies such as structured mentorship, leveraging institutional resources, and embedding research into clinical workflows can help overcome these challenges. By developing collaborative networks and building a culture that values inquiry, clinicians can contribute to impactful scholarship. Ultimately, these partnerships not only enhance clinical outcomes but also promote the growth of clinician-scientists who drive meaningful progress in healthcare. Together, we can create a sustainable model for clinician-driven research.
Corresponding Author
Adam P. Weaver
Connecticut Children’s
399 Farmington Ave
Farmington, CT 06030
Phone: (860)-966-2380
Fax: (860) 837-5649
aweaver@connecticutchildrens.org
Conflict of Interest
Adam Weaver and Lauren Butler have no disclosures. Christopher Kuenze serves as a paid consultant for Inova Health.