INTRODUCTION
Roughly 6% of the population reports Achilles tendon pain during their lifetime. Of this percentage, about one-third will have insertional Achilles tendinopathy (IAT).1 This condition is typically caused by an increase in mechanical loading of the Achilles tendon, particularly in a manner in which the tendon is loaded in extreme ranges of ankle dorsiflexion.2 Clinically, symptom presentation indicative of therapeutic intervention includes reports of retrocalcaneal pain, redness, swelling, tenderness to palpation, and gait impairments. This can lead to an abnormality of the posterosuperior part of the calcaneus where there is a bony enlargement at the attachment of the Achilles tendon, described as a Haglund’s deformity. This condition was first described in 1927 by Scandinavian orthopedics professor Patrik Haglund.3,4 The deformity itself can exist for years with normal function, but in cases when the development of the deformity occurs rapidly, and/or pain is present and function is impaired, conservative management is indicated. When these impairments coexist with the visible deformity, it is known as Haglund’s Syndrome.
Initial treatment focuses on identifying and managing pain generators—commonly retrocalcaneal and subcalcaneal bursitis or Achilles tendinopathy—all of which are subject to increased mechanical pressure from the Haglund’s deformity itself. Treatment approach includes physical therapy for examination and prescription of appropriate strengthening, stretching, manual techniques, and utilization of heel lifts and modalities. Due to the complexity and chronicity of this condition, conservative efforts take time. With an evidence-based approach, successful conservative management can take within 12-18 months. If symptomology does not improve given adequate time, surgical excision of the Haglund deformity may be an performed.4 The literature presents a wide-variety of surgical approaches and varied timelines in postoperative protocols. The purpose of this clinical commentary is to review current literature and propose a criterion-based protocol recommending early weight bearing when significant repair of the tendon is not necessary.
ANATOMY
The Achilles tendon is the strongest and thickest tendon of the human body.5 It originates from the aponeurosis of the gastrocnemius, soleus, and plantaris muscles and inserts 2 cm distal to the posterosuperior calcaneal prominence.1 These muscles work together as the primary plantar flexors of the ankle and the Achilles tendon is subject to tensile loads up to twelve times body weight during activities such as running and jumping.5,6 Degenerative changes within the Achilles tendon insertion, marked by loss of parallel collagen structure, loss of fiber integrity, fatty infiltration, and capillary proliferation are telltale signs of insertional Achilles tendinopathy.1
FINDINGS AND PATHOLOGY
Compression of the Achilles tendon insertion against the calcaneus may occur in extreme ranges of ankle dorsiflexion and is a risk factor for development of IAT.7 It is rare for the tendinopathy to result from compression in dorsiflexion alone, such as with stretching the ankle into dorsiflexion, but instead typically manifests itself from compression during the stretch-shortening cycle, such as when running up a hill.2
Insertional Achilles bone spurs, or osteophytes/enthesophytes, appear to be more common in those with IAT (65-80%) than in people without foot pain (25-35%). That said, many people may have the presence of posterosuperior calcaneal prominence, known as the Haglund’s deformity, but have little to no pain or dysfunction. Research has indicated that the size of the osteophytes, rather than the presence of the osteophytes may be the determinant in whether the patient experiences symptoms. Chimenti et al. found that bone spurs were significantly longer on the symptomatic side (12.9 mm) compared to the asymptomatic side (8.9 mm, p=0.01) and a control group (3.5mm, p=0.03).1 Therefore, it has been hypothesized that the pain producing source in patients with a Haglund’s deformity is the posterior-superior calcaneal prominence compressing on the Achilles tendon and retrocalcaneal bursa.1
Currently, there is no definitive etiology for Haglund’s Syndrome, however probable causes include high arch of the foot, tightness of the Achilles tendon, and heredity.4 It typically affects middle aged people, and is more common in females.4 In athletes, it is most common in runners and in individuals who wear compressive footwear such as skates or boots, including alpine and cross-country skiers, particularly in those who maintain a high annual training volume. Similarly, in non-athletic populations, tight or ill-fitting dress shoes often contribute to symptom development due to repetitive mechanical compression and friction at the heel.4,8
CLINICAL SIGNS AND IMAGING
Diagnosis of Haglund’s Syndrome is typically made based on the combination of the patient’s history and clinical findings. Commonly, patients describe pain localized to the retrocalcaneal region.8 The patients also display posterosuperior calcaneal prominence, and tenderness to palpation laterally and medially to the Achilles tendon at the level of the posterosuperior border of the calcaneus. This tenderness is often associated with redness, swelling and warmth. Pain may also be evoked with passive or active end-range ankle dorsiflexion,2,8 and is most notable as the patient begins to walk after periods of rest.4 Patients will likely demonstrate gait impairments due to pain. Often, pain with movement into extreme dorsiflexion, such as during calf raise/drop over a step is more painful than submaximal hopping in those with IAT.2
There are currently no clear cut radiologic criteria for early diagnosis, however, radiographic changes observed on lateral radiographs may assist in diagnosis.4 In the later stages of Haglund’s Syndrome, radiographs will depict increased bony prominence at the posterosuperior part of the calcaneal tuberosity, accompanied by calcaneal bursal swelling and increased density in the pre-Achilles bursae.4 In questionable cases, an MRI may be obtained which will depict posterosuperior calcaneal spurring with impingement on Achilles tendon.4 That said, it is important to be mindful that those with an asymptomatic pathology will be positive using this reference standard even though they do not have a painful variant of the condition, so it is important to factor in both the radiographic changes as well as the patient’s symptom presentation.2
CONSERVATIVE TREATMENT
Patients with Haglund’s Syndrome typically begin with conservative treatment in efforts to improve overall level of function and alleviate symptoms. Supported by a Grade A recommendation according to Dilger et al. exercise has the strongest evidence for reducing pain in this population.9 Specifically, strengthening programs designed to load the tendon are crucial in the rehabilitation process. Eccentric calf strengthening was previously considered the “gold standard” for treatment in Achilles tendinopathy; however, more recent evidence suggests that the type of muscle contraction (concentric vs. eccentric vs. isometric) is not as important as long as the load is heavy and slow.10–13 It is up to the discretion of the physical therapist to determine the appropriate amount of loading based on patient tolerance, but typically it is both safe and effective to begin loading with isometrics and progress according to tendon loading principles.13,14 Specific to IAT, patients achieve greater benefit with strengthening exercise when limiting the degree of ankle dorsiflexion, such as by placing a towel roll under the heel when performing a heel rise, as it prevents compression between the tendon and the calcaneus.1,9 That said, complete avoidance of ankle dorsiflexion may have a nocebo effect in prolonging recovery and may reduce outcomes.15 Therefore, a graded progression of ankle dorsiflexion as symptoms allow should be used during calf raise exercises in this population as displayed in Figure 1.2
With Grade B evidence, extracorporeal shockwave therapy (ESWT) has been cited in the literature as another option in reducing IAT pain, specifically when exercise has been unsuccessful.16 According to a systematic review performed by Zhi et al. the overall satisfaction rate with ESWT of 73.7% was much higher than that of eccentric exercise at 45.6%. That said, other evidence surrounding ESWT for treatment of IAT provides mixed outcomes.9 Some research points toward ESWT being more useful for short-term relief, while long-term benefit is no different than when compared to sham treatment.16–18
Consideration should also be given to sport-specific or daily movement patterns that involve excessive ranges of dorsiflexion during the stretch-shorten cycle, such as running backwards, changing directions, or running up steep inclines.2 Activity modification to reduce or modify these potentially symptom-provoking movements is the keystone of quality conservative management. Once symptoms are managed, building load and volume using objective criteria to measure progress guides the sport specific training and determines return to sport readiness. After these non-operative options have been employed and found to be unsuccessful, additional interventions may include stretching, footwear modification, anti-inflammatory medication, and local steroid injection.4 There is no “one-size-fits all” approach for footwear modifications; rather, the optimal strategy is the one that provides the patient with the most relief, as there is limited research guiding specific recommendations in this area. Typically, methods that work to offload the bony prominence and/or reduce friction on the symptomatic area are found to be beneficial. These strategies include, but are not limited to, the use of heel inserts, backless or loose-fitting shoes, footwear with an upper trim line positioned below the bony prominence, or adding internal heel padding.19 For professional athletes, the authors recommend that anytime the athlete is not training, they spend time in a backless shoe, sandal or clog. Further, for athletic footwear such as cross country (XC) ski boots, cycling shoes used for indoor training, or athletic walking shoes, removal of the entire posterior wall of the boot or shoe is recommended to eliminate compression, with replacement by a soft neoprene backing. Conservative efforts are typically trialed for anywhere from six months8,20 to 18 months,21 and have demonstrated high success rates with 85-95% of patients achieving symptom improvement.22 In cases where patients fail to experience meaningful symptom reduction within this timeframe, surgical intervention is generally considered.
SURGICAL TECHNIQUE
Surgical excision of the Haglund’s deformity frequently includes concomitant treatment of the insertional Achilles tendinopathy often present in this condition. The intra-operative evaluation of the health of the Achilles tendon will influence the surgeon’s technique, and as such the post-operative precautions. The most common approaches are endoscopic Achilles tendon debridement without suture fixation, open Achilles tendon debridement with suture repair, and open Achilles debridement re-attachment to the calcaneus with suture anchors. For severe cases involving significant tendon damage or chronic IAT, a different surgical approach is required via tendon augmentation with a flexor hallucis longus (FHL) tendon transfer. Further discussion of which is beyond the scope of this clinical commentary.
The first step of surgical intervention is Haglund’s removal with Achilles tendon debridement, which involves a lateral retraction or partial detachment/release of the Achilles tendon in order to expose the retrocalcaneal bursa.4 From there, the retrocalcaneal bursa is excised followed by the removal of the Haglund deformity. This procedure can be performed with either open or endoscopic approaches. Although requiring more skill from the surgeon compared to the open approach, the endoscopic approach offers lower complication rates (5.3% vs. 12.3%), less conservative rehabilitation process, and quicker return to daily activities (6-12 weeks vs. 6-36 weeks).8
The above approach is taken if 50% or less of the Achilles tendinous insertion is detached.23 If greater than 50% of the tendon is detached, an Achilles repair procedure utilizing anchors is necessary.23–25 Typically, patients who have undergone this procedure are treated with a short period of non-weight bearing with immobilization and weight bearing restrictions for up to six weeks.25
The second option utilizes a suture anchor construct. The suture anchor repair is a soft-tissue fixation which enables an hourglass pattern of ultra-high molecular weight polyethylene suture to be laid over the distal end of the tendon. This 4-anchor construct provides a knotless repair and a greater area of compression for the Achilles tendon on the calcaneus, improving stability such that immediate postoperative weightbearing and range of motion is possible.26 This option is performed when there is greater pathology of the tendon, thus requiring more significant debridement. Patients are non-weightbearing for two weeks after this procedure before they can begin a weightbearing progression.
A literature review (results displayed in Table 1) reveals large variance among rehabilitation guidelines. Some are more liberal and suggest immediate partial weight bearing,27 while some are more conservative and contraindicate weight bearing until the six week mark.8 Due to the lack of general consensus among outcome studies, this commentary will focus on postoperative protocol recommendations for healthy patients undergoing Haglund’s removal with endoscopic and open Achilles debridement, where tissue and surgical debridement or anchor placement are sufficient to allow early weightbearing.
POST-OPERATIVE REHABILITATION
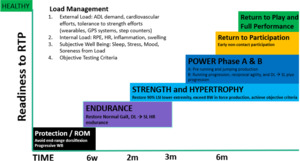
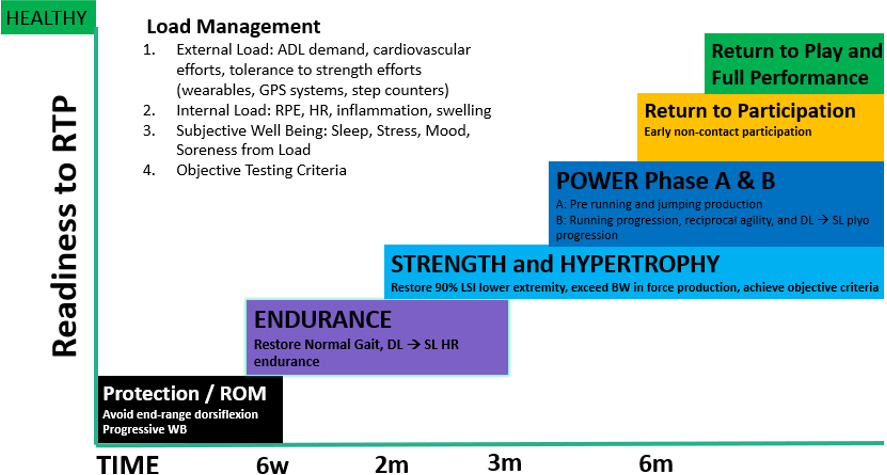
Following Haglund removal and Achilles debridement procedure, the utilization of an objective, criterion-based progression that aligns with physiological healing is essential for successful rehabilitation. The early protection phase emphasizes inflammation control, minimizing atrophy, and optimizing function. Subsequent phases are guided by tissue proliferation and tendon remodeling. Therefore, the following five-stage rehabilitation program is recommended for a safe progression toward return to sport. Figure 2 presents general timelines for the phases of rehabilitation.
Phase 1: Protection, ROM, Muscle Activation
The protection phase begins with a two-week non-weight bearing phase while the patient is in a splint. After two weeks, the splint is removed and the patient is placed in a postoperative boot. This boot is worn for weeks 2-6 while the patient can begin to bear weight as tolerated starting at week 3, progressing towards their pre-surgical daily step count. Though pain is expected, it should not exceed 3/10 on the VAS pain scale relative to per step tolerance or total volume attempted per day. During week 5-6, the patient may begin to wean out of boot and into a supportive shoe. If deemed beneficial, a heel lift may be placed in a supportive shoe while transitioning out of the boot. The patient is advised to only advance dorsiflexion beyond neutral if pain free for the first four weeks after surgery, as not to place excess stress on the debrided and re-anchored Achilles tendon.
Physical therapy goals of this phase include management of edema, restoration of both active and passive muscle recruitment, improving range of motion, and safe, pain modulated ambulation. The physical therapist must be astute in observing clinical indicators that ensure a favorable response to early weight bearing combined with prescribed exercise, and reduce load or exercise if swelling or pain in the area does not recover within 48 -72 hours.31
It is common for the patient to have developed compensatory patterns after months of pain and trialed conservative treatment; therefore, it is essential to focus on correcting compensatory patterns at this stage. This is addressed early with exercise to target foot intrinsics and arch doming. This is a critical skill when aiming to limit compensatory patterns in gait when transitioning from a walking boot to a shoe. Early recruitment of the gastroc-soleus complex with low-load plantarflexion-based exercises, such as band resisted plantarflexion or seated heel raises, is essential during this phase of rehabilitation to establish satisfactory muscular activation patterns. Early use of Neuromuscular Electrical Stimulation (NMES) with exercise can assist in establishing proper neuromuscular patterning and avoidance of compensatory patterns from deep posterior and lateral compartment musculature. Recommended postoperative NMES parameters for the calf mirror those used for quadriceps activation after knee surgery: 30–50 Hz frequency, 250–400 µs pulse duration, maximum tolerable intensity producing a visible contraction, and a 1:1–1:2 duty cycle (10 s on, 10–20 s off).32,33 Blood flow restriction (BFR) training may also be used two to three times per week to augment early exercise and challenge the gastroc-soleus complex without requiring a heavy external load that would be contraindicated at this stage of rehab. Recommended parameters include cuff placement proximal to the calf, an occlusion pressure of ~60-80% of limb occlusion pressure, low external load, and four sets per exercise with repetitions ranging from 15-30 reps per set with rest intervals of 30-60 seconds.34 Both with exercise and with functional activities, it is important to avoid extreme dorsiflexion ranges during this phase as to allow for appropriate healing.
Phase 2: Endurance
The endurance phase aims to restore functional, weight bearing ankle ROM in all directions while transitioning to a normal gait pattern in two shoes. A supportive, cushioned style shoe may be more comfortable for the patient at this stage of rehab. The footwear modifications described in the conservative treatment section may also be implemented to offload the sensitive incision. Mild swelling is typically common, and the patient may benefit from use of compression stockings during this phase. Manual techniques will be utilized if the patient does not demonstrate adequate midfoot, subtalar, and calcaneonavicular joint mobility necessary for correct gait patterns. It is also useful for the patient to track their daily step count, which allows adequate progression to their previous ADL and work demands without symptom exacerbation. The most consistent load stimulus early in this phase is walking, and achieving previous work and ADL demands is prudent before progressing strengthening.
The American College of Sports Medicine (ACSM) guidelines defined endurance exercise as high repetition (reps), 15+ reps, low load at a submaximal resistance with a rate of perceived exertion (RPE) of 7/10 at 40-60% of 1 rep max (RM). In conjunction with the physical therapist’s guidance, it is crucial that the patient balance exercise progressions with ADL advancements (increased weightbearing, step count), in order to optimize overall function. During this phase, post operative treatment strategies follow the same evidenced based practices in tendon loading as non-operative cases once the tissue healing time is observed. If concentric and eccentric plantarflexion and dorsiflexion is symptomatic, isometric loading in shortened and lengthened positions under appropriate load is low risk and will adequately prepare the patient for the ROM required to walk once in a shoe. This can be accomplished with a seated heel raise, or an isometric squat hold in a pain-free dorsiflexion range. If painful in a full weightbearing position, variations can be performed with a submaximal load over a 10 to 30 seconds isometric hold to achieve maximal fatigue.35 A symptom-guided, graded progression of ankle dorsiflexion range of motion may be initiated during this phase and can be used during lower extremity strengthening exercises.
Plantarflexion strength is critical to normal walking, which requires up to 3.3x bodyweight (BW), as such, achieving the ability to perform a single leg heel rise which requires ~3x BW, is arguably the most important endurance-based goal to achieve.6 Introducing pain free load in varied positions is prudent early in rehabilitation. Isometric or isokinetic plantar flexor strengthening can be initiated in this phase, starting in a resisted open chain with knee straight as well as seated heel plantarflexion (PF). The weighted seated single-leg heel-raise is a safe and early way to begin loading the calf muscle tendon unit (MTU) as this does not produce the same force as the standing heel raise.6 Once the patient can tolerate standing double limb and single limb PF, a gradual progression toward full dorsiflexion (DF) and PF with a calf raise can be utilized, such as depicted in Figure 1. Once a patient is able to perform 20 seated single-leg heel-raise repetitions at 50% bodyweight, they are more likely to have the capacity to perform a standing single-leg heel raise.36 Other objective measures for the patient to progress into the strengthening phase are found in Table 2.
Phase 3: Strength
Once the endurance criterion is achieved, the focus in rehabilitation is shifted towards the development of muscle hypertrophy (muscle cross sectional area) and strength (defined as maximum force exerted). This is arguably the most important phase of the patient’s recovery as it stimulates neuromuscular adaptation (uptake in EMG activity) in the calf MTU, and offsets atrophy that occurs in the first six weeks. The authors advise ample time devoted to the strengthening phase to set the athlete up for success in the power phases. Discerning the appropriate intensity of exercise in the hypertrophy phase is debated, but the key is encouraging complete fatigue of the calf MTU each set.38 It is suggested the patient targets 8-12 reps at 8-9/10 RPE or 40-60% of 1 RM or maximum voluntary isometric contraction (MVIC).
In general, a strength training stimulus represents an increase in weight and decrease in repetitions, with recommended sets of 1-5 reps > 60-70% of 1 RM or MVIC with RPE at 8-9/10. In this phase, the physical therapist should initiate heavy loading. As described in conservative management, the type of load (isometric, concentric, eccentric) is determined by the physical therapist, specific to the needs of the patient. Once determined, the key is that the load stimulus should target enough load to fully fatigue the calf muscle tendon unit (calf MTU) at the end of the repetition or set, guided by numerical pain rating scale proposed by Silbernagel et al.13 This progression is imperative in order to facilitate adequate tendon remodeling and neuromuscular adaptation.39 Achilles tendon pain and soreness should be monitored both during exercise and for the following 24-48 hours.31 Measurable strength gains are typically 10-15% every four to six weeks.40
It is essential that the patient spend sufficient time in the strength phase to build a solid foundation for success in future rehab phases. The author’s recommend maximizing the time devoted to this phase, best with multiple four to six week periodized training blocks. Figure 2 is often used as a visual aid throughout the rehab process to help patients understand the purpose and progression of each phase within the rehabilitation continuum. To advance into the next phase, the physical therapist should assess the criteria for progression outlined in Table 2. If the criteria are met, the patient may transition into Phase 4A: power and pre-agility.
Phases 4-5: Power, Plyometrics, Agility and Return to Sport
Phase 4A: Power Development prior to running and jumping
Phase 4B: Power, Running Progression, and Plyometrics
Phase 5: Return to Sport
Power Phase 4A emphasizes preparation for agility by integrating pace, power, and acceleration/deceleration training. The Achilles tendon undergoes loading equivalent to approximately three times body weight during stance in normal walking, and up to twelve times body weight during activities such as running.6 It is recommended that time is devoted in rehabilitation to pace and power prior to introducing impact. For example, progressing with an external cue of metronome rebounding double leg heel raises (2.5 x BW) can combine early endurance and stretch-shortening cycle stimulus.6 Similarly, focusing on acceleration with a high speed low step-up (>2.9x BW) and deceleration with drop squats or countermovement jump (3.4x BW) can also introduce this skill capacity but with far lower loads than actually jumping (4.8x BW for double leg hopping and 6.7x – 7.1xBW for single leg hopping and forward hopping). If the athlete benefits from a slower progression, these tasks can be introduced with lower bodyweight loads on a leg press or offloaded with high resistance exercise band (Figure 3). The other listed tests and measures in Table 2 can also be programmed into rehabilitation and progressed until the listed criteria are met. If available, isokinetic and power plate assessment tools can be utilized for assessment in this phase.
Once Phase 4A of the power criteria is met, the patient may transition into Phase 4B which consists of running and jumping to build maximal strength, reactive and explosive power for sport. The goal of this phase is to prepare the patient for the demands of the sport or activity they are aiming to return to. Like in the strength phase, it is recommended that the patient perform at least four to six weeks of progressive agility, power and plyometric training under the guidance of a physical therapist before the performance of the Phase 4B criteria for progression tests.
During this phase, it is important to carefully dose the number of touches and reps with the load volume equation (sets x reps x weight) to allow for adequate recovery time, especially as the patient should continue strength training at this time. Monitoring load progression is imperative to avoid increasing multiple variables simultaneously, particularly in efforts to reduce excessive mechanical stress and avoid tendon irritation. Introducing jumping can be offloaded with spring assistance of a thick resistance band to build confidence and offload the takeoff and landing demand in earlier phases. Plyometric activity should be progressed in a similar fashion to previously proposed exercises, beginning with double limb tasks, advancing to single limb activities, and eventually including multiple planes of movement depending on sport-specific demands.
Finally, once all Power Phase criteria are achieved, the patient may perform the recommended test battery for Phase 5: Return to Sport and Return to Performance,41 outlined in Table 2. This includes running, agility, power and hop testing, but is not an all-inclusive list of tests and measures. If other tests better mimic the activity demands required for a full return to sport, they should be included among the test battery. Overall, an individualized approach should be utilized in the decision-making process in determining whether or not an athlete is ready to return to sport.
CONCLUSION
Haglund’s Syndrome presents a complex challenge to both patients and clinicians alike due to multiple anatomic pain generators and its impact on function and quality of life. Patients advised to trial a comprehensive conservative rehabilitation approach through strengthening, ESWT, stretching and other methods in efforts of alleviating symptoms. If conservative care is unsuccessful and disability persists, surgical intervention is taken. A lack of consensus exists in the current literature regarding post-operative rehab protocols for this procedure, serving as a barrier to postoperative recovery. Therefore, this clinical commentary offers a structured rehabilitation framework following Haglund’s removal and Achilles debridement. It prioritizes early weight bearing suggestions for healthy athletes, minding early tissue healing timelines to protect surgical repair. Ongoing rehabilitation recommendations are guided by a criterion-based progression approach, allowing each step of the rehab process to be tailored specific to the patient’s individual progress. This can serve as a tool clinicians can use to facilitate a safe and efficient return to activity following an Achilles debridement and Haglund’s deformity removal.
Corresponding author:
Mark Dudek PT, DPT
Howard Head Sports Medicine, 180 S Frontage Rd W, Vail, CO 81657
mark.dudek@vailhealth.org
330-604-2206
Conflicts of Interest
The authors report no conflicts of interest.

_or_leg_press_(right).png)

_or_leg_press_(right).png)