INTRODUCTION
Dry needling (DN) is a treatment technique that has been increasingly utilized by physical therapists (PTs) to address musculoskeletal pain and dysfunction since the early 2000s.1 For the purposes of this consensus statement, DN is discussed within the context of neuromusculoskeletal dysfunction, with emphasis on its application to movement-related impairments, muscle performance deficits, and pain presentations commonly encountered in physical therapy practice. DN is a skilled intervention in which a clinician inserts a thin, solid filiform needle (without injectate) through the skin to stimulate myofascial trigger points and other muscular, connective, or neural tissues, with the goal of addressing neuromusculoskeletal pain and movement impairments. The American Physical Therapy Association (APTA) recognized DN as part of the physical therapist scope of practice in 2009. Despite its growing use, techniques and procedural parameters vary widely and lack standardization in research and across clinical settings.2,3
Evidence from both laboratory and clinical studies supports the effectiveness of DN.4 In fact the field of DN research has witnessed a significant growth since 2008 with a sustained annual rise in the number of publications.4 DN can modulate biochemical mediators involved in pain transmission and inflammation, including calcitonin gene-related peptide (CGRP), substance P, and various cytokines.3,5–8 A 2023 umbrella review by Chys et al. synthesized findings from 36 systematic reviews and concluded that DN is generally more effective than sham or no treatment and comparable to other physical therapy modalities for short-term pain relief in some conditions.9 This review found DN was particularly effective for treatment of neck pain, shoulder dysfunction, low back pain, and plantar fasciitis.9 However, its impact on physical function was inconsistent, likely due to variations in treatment protocols and outcome measures. Combining DN with other interventions, such as exercise or manual therapy, appeared to enhance outcomes.9 Despite promising short-term effects, the review highlighted limited and inconclusive evidence for long-term benefits, underscoring the need for standardized protocols and more rigorous longitudinal research.9
There is a clear need to reduce variability in DN procedures across both clinical practice and research. In this manuscript, “musculoskeletal dysfunction” refers to neuromusculoskeletal pain and movement-related impairments that limit a patient’s ability to perform functional tasks without pain or restriction, consistent with presentations commonly managed in outpatient orthopedic physical therapy practice. The purpose of this modified Delphi study was to develop consensus statements to guide clinicians and researchers in the application of DN for use in individuals with musculoskeletal dysfunction. Additionally, a decision tree was developed to organize key clinical decision-making parameters and guidelines for implementing DN as a therapeutic intervention.
METHODS
Study Participants
Panelists were identified using purposeful sampling by the two authors who initiated the consensus project based on predefined criteria for expertise in DN and orthopedic physical therapy practice. Panelists were intentionally selected to provide diversity of clinical perspective, by representing varied educational backgrounds, practice settings, and geographic regions across the United States. To be considered an expert, panelists were required to have at least three years of clinical experience in orthopedic or sports physical therapy, a minimum of three years performing dry needling, consistent clinical use of dry needling in practice (defined as routine integration into patient care), and recognized involvement in clinical education, research, or professional leadership within the field. The nine panelists were considered experts in either sports or orthopedic physical therapy, practicing across the United States. A summary of panelist characteristics, including years of clinical practice, years performing DN, and average weekly DN treatment volume, is provided in Table 1.
Study Design
This study utilized a modified Delphi technique, in accordance with the guidance on Conducting and Reporting Delphi Studies (CREDES),10 with the purpose of establishing consensus on the use of DN for individuals with musculoskeletal dysfunction. The process included informational input, attention to prevention of bias, interpretation and processing of results, and external validation.10 The methods used in this study were similar to those described by Martin et al.11 and Disantis et al.12 A priori consensus was defined as ≥80% agreement among panelists, consistent with commonly accepted Delphi standards.
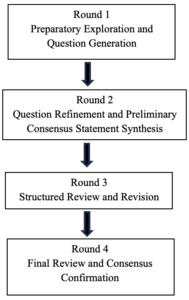
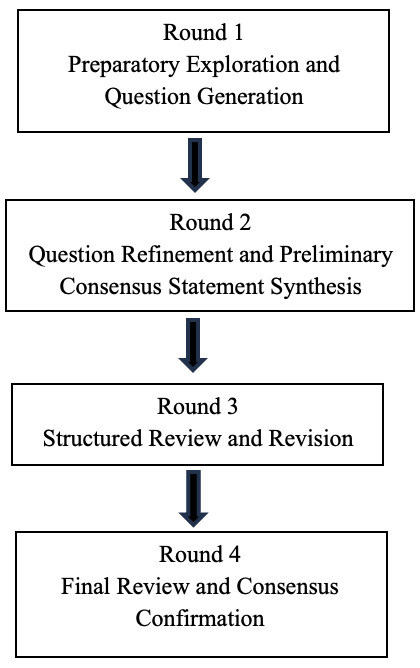
The consensus development occurred over four iterative rounds involving the panelists (Figure 1) as follows:
Round 1: Panelists were presented with an initial comprehensive set of questions outlining the purpose of the Delphi study and general questions developed by the senior author to begin framing the consensus statements. During this round, conducted via a virtual meeting, panelists provided open-ended feedback and suggested additions or revisions. The senior author synthesized this input into a refined set of specific questions for further evaluation. At the end of Round 1, panelists voted on the initial list of general questions to be addressed.
Round 2: In the second meeting, conducted via a virtual session, panelists reviewed and voted on the revised questions, contributed to the development of preliminary responses, and discussed the conceptual structure for a decision tree. Two authors consolidated the panel’s input to draft initial consensus statements, discussion points, and a preliminary decision-tree framework.
Round 3: The purpose of this round was to facilitate primarily editorial review and refinement of the materials. During this round, conducted via a virtual session, panelists participated in a focused review of the draft questions developed in Round 2, consensus statements, and decision tree, offering feedback to further clarify areas of agreement and divergence. The revised materials were then circulated for asynchronous evaluation, allowing panelists to independently provide written comments, ratings, and suggested modifications. All input from this combined iterative process was synthesized by the senior author and incorporated into the final set of revisions.
Round 4: In the final round, panelists reviewed the updated materials through online documents and provided final edits or approval. This round served as the confirmation phase typical of Delphi studies.
RESULTS
After the first round, the panelists agreed (9/9) that the following general questions needed to be answered:
-
What should determine a patient’s appropriateness for DN treatment?
-
What factors should be used to help guide the number of needles, anatomical regions treated, and overall treatment volume, as well as the aggressiveness of DN treatment?
-
When should treatment be directed at spinal segments?
-
What should guide the treatment of the extremities?
-
What are important post-DN treatment considerations?
After the second round, the panelists agreed (9/9) on the refined questions that would be used to develop consensus statements and discussion points to ultimately drive the decision tree as follows:
-
What clinical criteria should be used to determine a patient’s appropriateness for dry needling (DN) intervention?
-
What factors should be used to help guide the number of needles, anatomical regions treated, and overall treatment volume, as well as the aggressiveness of DN treatment?
-
Should movement quality (movement exam) be assessed before and after DN?
-
Under what clinical circumstances should initial DN treatment be directed at the spine?
-
What assessment findings should guide dry needling treatment for peripheral structures?
-
What key post-treatment strategies should be considered following DN to support outcomes?
Consensus statements, discussion points, and the decision tree (Figures 2A and 2B) received unanimous (9/9) agreement from the panelists following the fourth round. Figure 3 provides the key for the graphic figures.
CONSENSUS STATEMENTS AND DISCUSSION
Question 1: What clinical criteria should be used to determine a patient’s appropriateness for DN intervention?
Consensus Statement
Individuals with musculoskeletal-related pain, mobility deficits, muscle performance limitations, and movement dysfunction should be considered potential candidates for DN after being evaluated for absolute contraindications, precautions, and the need for treatment modifications or deferral.
Failure to adequately screen patients can increase the risk of adverse events, compromise patient trust, and undermine the intended therapeutic benefits of DN.13 The expert panel agreed that absolute contraindications include, but are not limited to the following: 1) inability to provide informed consent due to age, cognitive impairment, or communication barriers; 2) first trimester of pregnancy and pregnancies deemed “high risk”; 3) severe needle phobia; 4) active systemic or local infection; 4) open wounds at the treatment site; 5) tumors or cysts at the treatment site; 6) within the first six weeks following surgery: and 7) significant bleeding risk. Related to bleeding risk, International Normalized Ratio (INR) can be used to determine if someone is outside of normal ranges, though current evidence suggests that antithrombotic medication use alone should not be considered an absolute contraindication when appropriate precautions are implemented and individual bleeding risk is thoroughly assessed.14
Based on expert consensus and clinical precaution rather than definitive evidence, DN is typically avoided within the first six weeks following surgery. Local tissues surrounding surgical sites are generally avoided for up to twelve weeks postoperatively due to tissue healing considerations and potential infection risk, unless otherwise specified by a physician’s written order.
It should also be noted that unless the clinician has appropriate training, DN should avoid high-risk anatomical regions, such as the thorax, pelvic floor, neurovascular bundles, temporomandibular joint, and anterior neck. Based on expert consensus and established clinical safety principles, electrical stimulation in conjunction with DN should be avoided in proximity to pacemakers or other implantable cardiac devices due to the potential risk of device interference.
Precautions for DN include, but are not limited to the following: 1) immunosuppression; 2) children under 12 years old; 3) a history of psychological trauma or needle-related anxiety; 4) tissue fragility associated with elderly patients or corticosteroid use, 5) metal allergies such as nickel sensitivity; 6) needling in areas with dense neurovascular structures; and 7) atypical or sensitized pain presentations including widespread pain, allodynia, hyperalgesia, neuropathic pain, central sensitization, or complex regional pain syndrome. When precautions are present, clinicians must conduct a thorough risk-benefit analysis, consider modifications to technique, dosage, or needle selection, obtain additional medical clearance when appropriate, and document their rationale for proceeding with or deferring treatment. The identification of appropriate candidates requires clinical expertise, comprehensive assessment, and ongoing evaluation of patient response to ensure safe and effective treatment outcomes.
Question 2: What factors should guide the scope, intensity, and aggressiveness of DN treatment?
Consensus Statement
A structured approach to DN dosage can be estimated based on level of irritability and estimated treatment tolerance within the following potential clinical profiles: 1) high irritability with low treatment tolerance, 2) moderate irritability with moderate tolerance, and 3) low irritability with high tolerance.
It should be noted that significant standardization gaps exist in the current literature regarding specific dosage parameters and these recommendations represent expert consensus.6 The framework proposed here helps clinicians guide needle application strategy (e.g., tissue selection, distribution, and treatment extent), which tissues to target, and what dose of pistoning or electrical stimulation is appropriate. Effective decision-making balances a patient’s reported symptoms with a thorough assessment of movement dysfunction, neuromuscular patterns, and relevant impairments.
Consistent with panel consensus, the number of needles used, anatomical regions treated, overall treatment volume, and intensity of DN should be guided by clinical findings, patient presentation, and treatment goals. Three options are presented relative to irritability and tolerance.
High Irritability, Low Treatment Tolerance
In high-irritability presentations (pain >7/10), DN should be conservative and focused on calming symptoms. This includes a limited number of needles, minimal mechanical stimulation (1-2 pistoning passes or until first twitch response, whichever comes first). For the purposes of this consensus statement, a “piston pass” is defined as a single controlled advancement and withdrawal of the needle through the target tissue during DN.
In this context, treatment intensity is defined by both mechanical stimulation (e.g., number of pistoning passes and elicitation of local twitch responses) and electrical stimulation parameters (e.g., frequency and amplitude set at levels tolerated by the patient without exacerbation of symptoms). The primary goal of treatment is symptom reduction; therefore, careful consideration must be given to tissue selection to avoid provoking hyperirritable structures. For example, a patient with Complex Regional Pain Syndrome (CRPS) should not receive direct treatment in the painful area but rather target proximal tissues that may reduce regional sensitivity, such as the multifidi at spinal levels corresponding to the innervation of the sensitized region (e.g., lumbar L3 multifidus for knee pain presentations). Treatment should focus on tissues neurologically or mechanistically related to the pain source, recognizing that symptom origin may differ from the location of reported pain, as seen in nociplastic pain conditions, regional hypersensitivity states, and acute injury presentations.11,12,15
Moderate Irritability, Moderate Treatment Tolerance
In moderate-irritability presentations (pain 4-7/10), a regional or multi-site treatment approach may be appropriate. This involves treating both central and peripheral contributors with moderate pistoning (3–5 pistoning passes or until multiple local twitch responses are elicited per targeted tissue, adjusted based on tissue response and treatment goals) and applying electrical stimulation at a tolerable intensity that produces a visible or palpable contraction. In this context, treatment intensity, as previously defined, should be adjusted based on patient tolerance and clinical response.
For example, a patient with shoulder impingement may receive treatment to both the affected rotator cuff muscles peripherally and the cervical multifidus centrally at C5-C6 levels that correspond to the shoulder’s neural innervation, addressing both local tissue dysfunction and potential central contributions to the pain experience.
Low Irritability, High Treatment Tolerance
In low-irritability presentations (pain 0-3/10), a more comprehensive strategy may be warranted. This includes needling multiple regions with higher pistoning volumes (10-20 passes or multiple twitch responses per targeted tissue, adjusted based on tissue response and treatment goals) and applying electrical stimulation that produces a visible or palpable contraction. For example, this comprehensive approach may utilize multi-site electrical stimulation across the posterior chain, targeting the gluteus medius, gastrocnemius, hamstrings, L5–S1 multifidus, latissimus dorsi, and lumbar paraspinal musculature (e.g., erector spinae group) to address extension patterns and posterior trunk and lumbopelvic stability. Alternatively, a multi-site anterior chain approach may focus on the rectus abdominis, adductor longus, iliacus, rectus femoris, and tensor fascia latae to improve flexion control and anterior trunk and lumbopelvic control.
However, low-irritability and high-tolerance patients may not require treating all involved areas in a single session; treatment decisions must be guided by objective examination findings, recognizing that more DN does not equate to better or more effective outcomes.
Question 3: Should movement quality (movement-based examination) be assessed before and after DN?
Consensus Statement
A thorough functional movement examination should be performed on all patients before and after DN intervention to assess pain limitations, movement asymmetries, altered segmental timing, and motor control deficits.
A movement-based assessment is a foundational element of clinical reasoning and a key component of clinical decision-making when considering DN intervention. Examples of these include, but are not limited to, components of the Selective Functional Movement Assessment (SFMA), Functional Movement Screen (FMS), Overhead Squat Assessment (OHS), Single Leg Squat Test, and others. These movement-based assessments, in conjunction with patient history, symptom behavior, and standard clinical screening, help determine patient appropriateness for DN intervention by identifying contraindications, precautions, and treatment tolerance, guide tissue selection (assist in informing decisions between central (spinal) and peripheral treatment targets), and establish a functional baseline for measuring treatment outcomes.
Movement should be evaluated in the context of the patient’s activity demands and regional interdependence, recognizing that impairments distant from the site of pain may contribute to the primary complaint.11,12 Screening often reveals compensatory patterns such as asymmetries, altered segmental timing, or motor control deficits that are not evident in localized examinations.16,17 Identifying these patterns is essential for targeting relevant central or peripheral regions rather than focusing solely on the symptomatic area.11,12 Movement assessment also creates an objective feedback loop by allowing the clinician to monitor functional changes in motor control, range of motion, or protective strategies pre- and post-intervention.16,18 Furthermore, it can expose latent movement deficits, defined as impairments in movement quality, control, or coordination that may not be apparent during low-demand tasks but emerge during higher-load, speed, or complexity-based activities, as described in injury risk profiling literature.17 As such, DN should be viewed as one component within a broader movement-focused treatment plan, rather than a stand-alone solution.
In addition to evaluating movement, clinicians must also consider psychosocial and lifestyle-related factors that may influence movement, irritability, and tissue response. These factors include, but are not limited to, things such as nutrition, hydration, sleep quality, psychological stress, pain during activity, and history of previous injury. These factors should be considered within the broader context of associated movement impairments contributing to the patient’s chief complaint.11 Failing to address these contributors may limit treatment effectiveness or lead to recurrent movement dysfunction. These variables should be captured during the subjective examination and integrated into both the assessment and plan of care. Educating patients on how these factors relate to their pain and performance enables them to take an active role in their recovery and may increase the likelihood of long-term improvement.
Ultimately, movement-based testing as well as psychosocial and lifestyle screening provide the context in which DN can be most impactful. Targeting not just the symptoms but the contributing behaviors, patterns, and impairments allows clinicians to be better equipped to provide effective, patient-centered interventions that create lasting change.
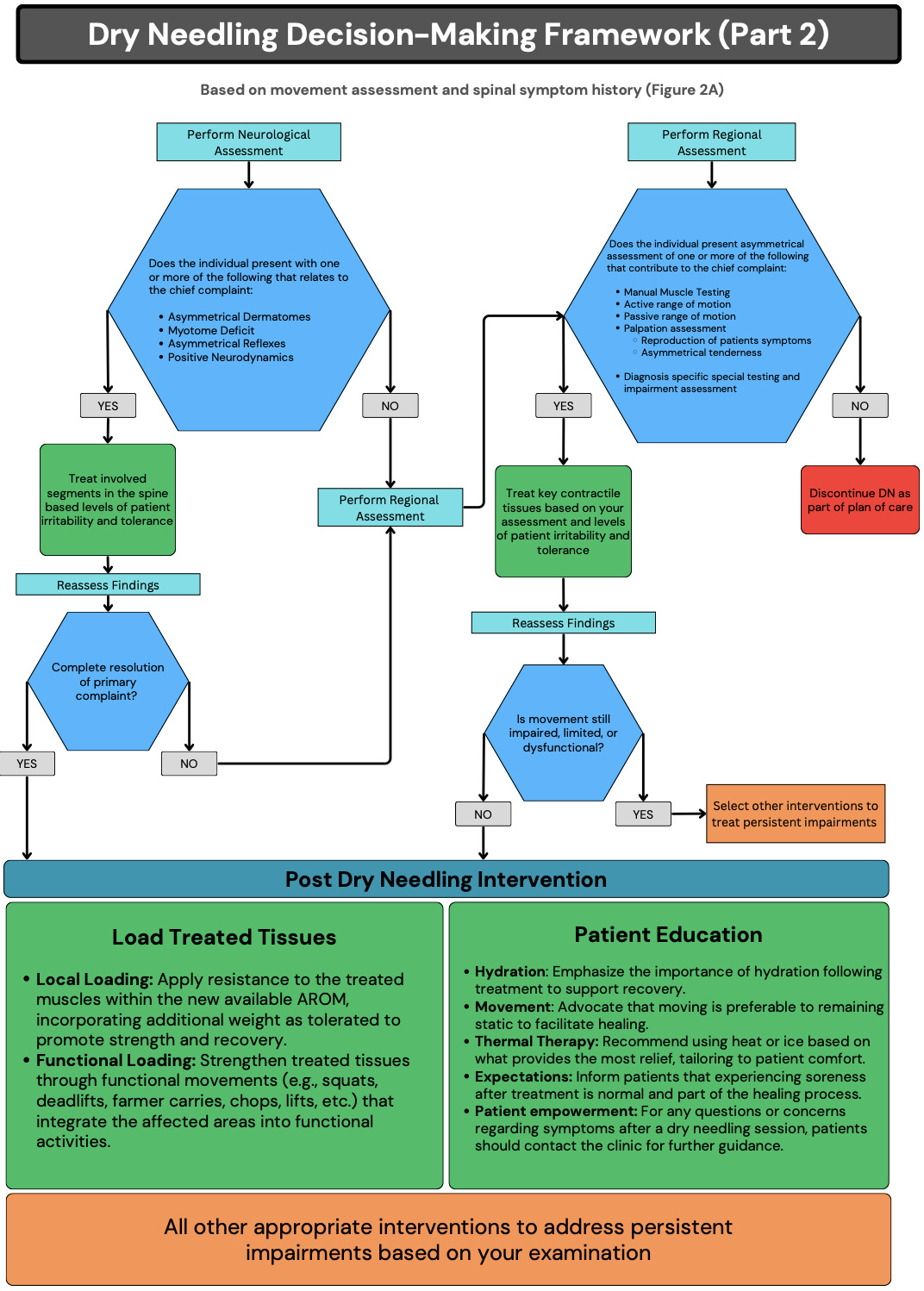
Question 4: Under what clinical circumstances should initial DN treatment be directed at the spine?
Consensus Statement
DN directed at the spine should be considered when neurological assessment reveals deficits in dermatomes, myotomes, deep tendon reflexes (DTRs), or neurodynamic mobility.
Neurological assessment findings (including structured testing of dermatomes, myotomes, DTRs, and neurodynamic mobility) are referenced in this consensus statement to establish clinical decision-making standards and reassessment benchmarks, rather than to instruct clinicians on exact examination techniques. A thorough neurological assessment is essential for determining the appropriateness, safety, and clinical prioritization of DN. The clinician should be aware that symptoms may originate from central sensitization or spinal segment dysfunction even in the absence of overt spinal complaints. Neurological assessment should be performed regardless of whether the primary complaint involves the spine or extremities. Neurological assessment is essential in differentiating between central and peripheral drivers of dysfunction and will also help inform the clinician in the appropriateness of spinal segmental treatment, dosage decisions, and post-treatment reassessment.19–21
Because dermatomal assessment is inconsistently operationalized in musculoskeletal literature, a brief description is provided to clarify how sensory findings are interpreted within this consensus framework. Dermatomal sensory testing is conducted bilaterally using consistent mechanical stimuli (pinwheel, hot/cold, two-point discrimination).22,23 Findings of hypoesthesia, hyperesthesia, or allodynia may indicate spinal nerve root-level involvement or central sensitization.22,24 Tools like the Wartenberg pinwheel have demonstrated enhanced sensitivity in detecting subtle sensory changes across both cervical and lumbosacral distributions.23,25 Visually observed trophic changes (e.g., alterations in skin texture, color, or hair distribution) may indicate underlying neurological involvement and should be considered as part of the dermatomal evaluation.15,26 When dermatomal abnormalities are identified, these findings may support consideration of segmental contributions to peripheral symptoms and inform prioritization of spinal-directed DN, as well as serve as objective reassessment markers following intervention.
Myotomal testing provides insight into spinal segment integrity by assessing motor output from individual nerve roots.19,27 Because the orthopedic literature does not consistently distinguish between traditional manual muscle testing and myotomal assessment focused on segmental motor quality, brief operational descriptions are provided to ensure clarity, reproducibility, and consistency in how neurological findings are interpreted within this consensus framework. Clinical specificity in determining myotomal dysfunction improves when evaluating at least two muscles innervated by the same spinal segment but different peripheral nerves.27 For example, C6 may be assessed using the biceps (musculocutaneous nerve) and wrist extensors (radial nerve), while L3 can be evaluated via the quadriceps (femoral nerve) and adductors (obturator nerve). Testing should involve submaximal eccentric resistance held for 5–10 seconds, using the cue “Hold—don’t let me move you,” with an emphasis on evaluating recruitment quality, endurance, and fatigue rather than maximal strength. A positive test may present with the inability to generate or maintain a smooth, coordinated contraction. Deficits identified in two muscles innervated by different peripheral nerves but sharing the same spinal segment suggest a spinal segmental (central) driver, even in the absence of overt spinal symptoms.19,28 Bilateral comparisons and testing across multiple muscles within the same segment further assist with diagnostic specificity and allow for reassessment following intervention.19
DTRs should be tested bilaterally in both the upper (e.g., biceps, triceps) and lower extremities (e.g., patellar, Achilles). Clinicians should assess for symmetry, amplitude, and latency. Although individual variability exists, asymmetric or diminished reflexes may suggest segmental motor pathway involvement.19,28
Neurodynamic testing evaluates the mechanical sensitivity and mobility of the nervous system and should incorporate structural differentiation to confirm neural involvement.29 Whether the central nervous system is the driver for peripheral nerve dysfunction will depend on other objective findings. In the lower extremity, tests such as the Straight Leg Raise (SLR), Femoral Nerve Tension Test (FNTT), and Slump Test assess mechanosensitivity of the sciatic and femoral nerves and associated spinal segments.28,29 In the upper extremity, Upper Limb Neurodynamic Tests (ULNTs) are used to evaluate the median, ulnar, and radial nerves.29 A test is considered positive when symptoms are reproduced during movement and are subsequently modified with structural differentiation—defined as the use of remote movements to selectively load or unload the nervous system—indicating possible neural involvement.30
When neurodynamic testing indicates peripheral nerve involvement without radicular findings (e.g., negative myotomal, dermatomal, or DTR changes), treatment should address both peripheral and segmental contributors. Peripheral nociceptive input can trigger altered spinal processing where dorsal horn neurons amplify incoming signals, causing normally subthreshold inputs to become suprathreshold.15,31 For example, positive ULNT1 findings without C6–C7 radicular signs warrant assessment of peripheral entrapment sites (e.g., carpal tunnel, pronator teres) and consideration of segmental treatment at C6–C7 with the goal of modulating spinal segmental contributions to the patient’s symptoms. This dual approach recognizes that peripheral nerve sensitivity may reflect both local mechanical factors and contributions from central neuroplastic mechanisms at the segmental level.30
Question 5: What assessment findings should guide dry needling treatment for peripheral structures?
Consensus Statement
DN should be directed at peripheral structures when impairments are identified through manual muscle testing, range of motion assessment, flexibility evaluation, pain provocation testing, and diagnosis-specific special tests.
Dry needling directed at peripheral structures is intended to modulate local nociceptive input, improve neuromuscular function, and restore movement capacity when regional impairments such as strength deficits, range-of-motion limitations, flexibility restrictions, or pain reproduction are identified during examination. A regional assessment is essential for identifying segmentally and functionally relevant asymmetries that contribute to the patient’s primary complaint. This assessment guides DN decision-making by localizing dysfunction and differentiating among neural, stability or motor control, mobility, myofascial, and articular impairments. Regional examination complements neurological findings by identifying locally expressed impairments that may be influenced by segmental or central factors.
Potential key tissues and structures contributing to the patient’s impairment can be confirmed through targeted palpation. This palpation focuses on identifying areas of muscle dysfunction that correlate with movement deficits found during regional assessment. While myofascial trigger points—defined as hyperirritable nodules within a taut band of skeletal muscle—may be identified, limited classification reliability of trigger points exists, supporting a focus on broader dysfunctional muscle regions that contribute to functional limitations.32 Palpation should assess overall muscle tone, tissue quality, and areas of tenderness that reproduce symptoms relevant to the patient’s complaint.
Treatment decisions should prioritize functional relevance rather than isolated trigger point identification. Within the proposed decision-making framework, palpation serves as a confirmatory assessment to localize peripheral tissues contributing to identified movement impairments. Effective palpation begins by locating the target muscle and assessing tone along and across fiber directions to identify regions of altered tissue quality or increased tension that correspond with deficits observed during regional and movement-based examination. Rather than systematically searching for specific trigger point classifications (e.g., active, latent, satellite, or central), clinicians should focus on tissues that reproduce familiar symptoms or disrupt functional movement patterns. Reproduction of symptoms during palpation—particularly when linked to task-specific limitations—supports the clinical relevance of the identified tissue.33 When such dysfunctional tissue is confirmed, dry needling may be applied using techniques scaled to the patient’s irritability and treatment tolerance, consistent with the dosage framework outlined in the decision tree, to support restoration of neuromuscular function and movement quality.34
Question 6: What key post-treatment strategies should be considered following DN to support outcomes?
Consensus Statement
Immediately after DN treatment and re-assessment, patient goal-centered therapeutic exercises should be performed to load the treated tissue and support efficient recruitment patterns and movement quality. Education on post-treatment expectations should also be provided.
The effectiveness of DN may extend beyond the intervention itself, with post-treatment strategies serving as the critical bridge between immediate physiological changes and long-term functional outcomes. Although DN may produce immediate improvements in tissue mobility, pain, and neuromuscular function—defined here as the integrated interaction between the nervous system and musculoskeletal tissues governing movement coordination, force production, and motor control—these effects require intentional post-treatment reinforcement to maximize therapeutic benefit and support sustained recovery.
Exercises should reflect the patient’s goals and be guided by impairments identified during evaluation. Light to progressive loading may support motor control, reinforce efficient recruitment patterns, improve blood flow, and gradually restore movement quality.35,36 Research demonstrates that DN creates immediate improvements in neuromuscular efficiency,37 requiring less motor unit recruitment to achieve greater force production, supporting the use of immediate motor pattern reinforcement exercises to capitalize on improved neuromuscular function.
In some cases, adjunct strategies such as blood flow restriction (BFR) may be used following DN to support early loading of treated tissues, particularly when higher-load exercise is not yet tolerated.38,39 In this context, BFR may assist with neuromuscular activation and allow for strength adaptations at lower mechanical loads, supporting the transition from immediate post-treatment changes to progressive loading.38,39 Other supportive strategies may include the use of heat or cold based on post-DN tissue response (e.g., soreness, stiffness, or localized sensitivity) and the need to support recovery and tolerance to progressive loading.
Movement-centered patient education supports recovery by promoting autonomy and establishing clear expectations for post-treatment activity and progression. Core messages may include engaging in appropriate movement to reduce post-treatment soreness, maintaining activity within tolerance to support progressive tissue loading, and understanding that mild post-treatment discomfort is common and typically resolves within 72 hours.
Clear communication regarding expected responses and follow-up is also recommended. Patients should be encouraged to monitor their response and maintain communication with the treating clinician as needed, supporting shared decision-making and ongoing progression of care.
Limitations and Future Directions
This consensus statement reflects expert opinion derived from a purposively sampled panel of experienced clinicians and should be interpreted within that context. While consensus was achieved across all statements, these recommendations do not replace the need for high-quality research to further clarify optimal DN dosage, tissue selection, and long-term outcomes. Notably, significant variability persists in the literature regarding DN parameters, the use of outcome measures, and treatment sequencing.
Future research should aim to investigate the mechanisms underlying DN effects, explore dose-response relationships, and evaluate how DN integrates with movement-based and rehabilitative interventions across different patient populations. Longitudinal studies examining functional outcomes and durability of effects are particularly needed to inform best practices.
CONCLUSION
This modified Delphi study established expert consensus regarding a structured framework to guide clinical decision-making in the application of dry needling for individuals with neuromusculoskeletal dysfunction. The resulting model organizes key domains—including patient presentation, irritability, movement assessment, neurological findings, and treatment tolerance—into a cohesive structure to assist clinicians in selecting and progressing interventions.
This framework is intended to be applied following a comprehensive subjective and physical examination and should be used to support, not replace, clinical reasoning. Rather than prescribing specific techniques or dosing parameters, it provides a structured approach to interpreting clinical findings and identifying functionally relevant treatment targets.
A central contribution of this work is the emphasis on integrating dry needling within a broader, movement-based rehabilitation strategy. Outcomes are not driven by dry needling in isolation, but by how it is applied in context, the patient reassessed, and function reinforced through appropriate loading, motor control, and patient education. By establishing shared terminology and decision-making structure, this consensus may improve consistency in clinical application and provide a foundation for more standardized research. Future research is needed to further investigate optimal dosing strategies, treatment sequencing, and long-term functional outcomes associated with dry needling within integrated rehabilitation models.
Conflict of Interest Statement
Jon Beougher is the Select Medical National Coordinator for Dry Needling Services and teaches dry needling courses for Select Medical. Kelby Church teaches dry needling courses for Evidence In Motion’s Functional Dry Needling program.Gray Cook receives consulting fees and is in a fiduciary role for Functional Movement Systems. Susan Falsone is the owner of Structure & Function Education, which provides dry needling continuing education. Tyler Waterway and Edo Zylstra are co-founders of Tactical Physio, which provides dry needling continuing education, and both teach dry needling courses through this platform.
.png)
.png)