BACKGROUND AND PURPOSE
Injury to the anterior cruciate ligament (ACL) causes an immediate functional disparity to knee joint proprioception and the central and peripheral nervous systems. The neurological and physical alteration in joint mechanics warrants targeted interventions to regain functional performance.1–3 Clinicians rely on progressive physical and neurological best practice intervention techniques to advance and guide rehabilitation and functional performance that leads to return to play sport parameters.3,4 After injury or surgery clinicians are presented with the challenge to improve or correct mechanical dysfunctions including bilateral and/or ipsilateral asymmetries in balance, strength, coordination, and reacquisition of skills. Recent data indicates the changes that occur in musculoskeletal or physical performance during the rehabilitation process may be ill-proportioned to the improvements noted by the central and peripheral neurological systems following ACL rupture and repair.1,3
Immediately post-injury there are drastic changes to the both afferent (sensory) and efferent (motor) components of the peripheral nervous system which results in disrupted proprioceptive and motor function.1,3 Activity at the brain and central nervous system (CNS) are therefore impacted by deficits in the neurophysiological somatosensory, motor excitability, and neural connective pathways throughout the body. The ligament rupture results in an altered feedback loop and changes of CNS processing of somatosensory information and concomitant alterations in motor responses.1,3 Patients struggle to accurately maintain kinesthetic awareness, joint position awareness, the accuracy of the velocity of movements, and muscle forces necessary to function appropriately.3 As a result, recent literature supports the use of treatment protocols that target somatosensory processing in the CNS in tandem with the management of impairments of pain, swelling, joint instability, and motor dysfunction.1,5 The impaired feedback from ligament rupture results in advanced stages of disaffirmations and degradative neuroplastic modifications that result in insufficient efferent motor responses, and lead to functional limitations related to lack of body/limb coordination, postural control issues, and gait deviations.3,4,6 For these reasons, researchers and clinicians attempt to target neural adaptation of both the peripheral and central nervous systems.3 Neural adaptation following a significant alteration in afferent activity is well documented in the literature for joint replacement where numerous afferents are lost and other systems adapt to provide proprioception-like afferents.3,4 While acute neural changes have been monitored in the brain during functional activities, there is very little empirical real-time evidence of central/brain neural modifications sequentially throughout the progressions of a post-ACL repair/rehabilitation program. Thus, monitoring brain and central nervous system performance throughout a rehabilitation program may provide objective metrics to improve the neuromuscular training and neurophysiological development needed during rehabilitation following an injury, such as a ruptured ACL.4,5
Quantitative electroencephalography (qEEG), commonly referred to as “Brain Mapping” is the utilization of a digitized signal to analyze changes in brain function and performance. Measuring feedback loops between sensorimotor stimuli and functional tasks allows clinicians to measure cortical electrical brain wave activity. These measures can be used to evaluate a variety of conditions thought to be rooted to the brain, such as, post-traumatic stress disorder (PTSD), attention disorders, autism, epilepsy, seizure disorders, dementia, headaches, traumatic brain injuries, sleep disorders and learning disorders.4,7,8 qEEG applies sophisticated mathematical and statistical analysis to the brain wave patterns and compares these observations to an universal database compiled of numerous healthy controls representing a variety of ages and genders.7 The qEEG system used in this study has added technological advances that allow for dynamic activities to be performed during the qEEG data collection, resulting in a more profound assessment of qEEG and neurophysiological metrics as part of a rehabilitation program for an ACL rupture. Such measures display real-time brain electrical activity that occurs during physical activities. The Cognionics (CGX) Quick- 20r v2, 21 channel state of the art dry EEG headset (CGX EEG) from CLR Neurosthenics, Inc (Manhattan Beach, CA), with LED impedance indicators is used to collect continuous electrical signals from the brain that measure the magnitude and timing of cortical activity across 19 active channels (with 2 reference channels) relative to a stimulus or a motor task.9 Cortical activity can be evaluated in response to a stimulus or functional task associated with the specific action.8
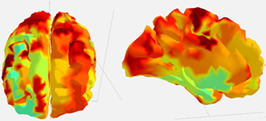
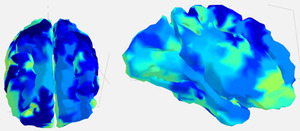
A well-functioning and active brain is classified as being in a regulated ready state with efficient brain connectivity as indicated by the red/orange highlights on a qEEG brain image.7,9 Two brain maps are presented in Figures 1a and 1b. Figure 1a represents a well-regulated state with little inhibitory drive which indicates efficient brain connections.7 Figure 1b represents an inhibited neurophysiological state indicating poor efficiency of brain connectivity. The image in Figure 1b was obtained within 24 hours of knee post-injury. The blue tints seen in Figure 1b are indicative of a dysregulated and poor electrical active brain state. The type and strength of the electrical impulses or brain waves can be analyzed to determine how different parts of the brain function and communicate during a given task. For example, different EEG signals: delta (0.5–4 Hz), theta (4–7 Hz), and alpha (8–12 Hz) waves have multi-faceted cortical sensory integrations used to promote motor activity that can reflect a motor and/or sensory activity period.10 Memory tasks are associated strictly with theta and alpha waves typically involved with inhibition or deactivation of sensorimotor activities.8,10 Evaluating the strength in electrical activity and band frequency of the brain waves throughout the cortex can quantitatively reflect the activity associated with a physical task.10
There are limited data identifying the cortical changes that occur throughout the linear progression of an ACLR rehabilitation program. Documenting the electrophysiological changes and accommodations at the brain and/or central nervous system level while progressing cognitive and physiological motor tasks used post-ACL injury or surgery may be valuable in developing more precise exercise progressions and return-to-play decisions. The purpose of this case report was to investigate the innovative use of qEEG and neurophysiological metrics to monitor the longitudinal and simultaneous change in brain and central nervous systems activity while measuring musculoskeletal function during an anterior cruciate ligament repair rehabilitation.
CASE DESCRIPTION
Subject Demographics
A 19-year-old, right-handed, Division I NCAA female lacrosse midfielder with 10 years of playing experience suffered a full thickness mid-substance anterior cruciate ligament rupture and a vertical posterior horn tear of the lateral meniscus near the intercondylar notch of the right knee. Clinical evaluation revealed moderate capsular swelling and a dysfunctional limb with pain during gait. Joint laxity following a Lachman’s anterior drawer, pivot shift, valgus, and varus stability tests revealed excessive anterior tibial translation and a grade 1 MCL and LCL sprain. The injury occurred on 01/24/2022, and findings were confirmed the same day via MRI. An arthroscopy with anterior cruciate ligament reconstruction using hamstring tendon autograft and a 5% lateral meniscectomy was performed on 01/28/2022. The subject volunteered to take part in the case study and signed an informed consent regarding patient confidentiality and the protection offered by the U.S. Health Insurance Portability and Accountability Act (HIPAA). This project received no outside funding and the research was completed by the University of Cincinnati Sport Science Research Team.
During the course of the rehabilitation program, the patient’s central nervous system, brain performance and musculoskeletal function were monitored longitudinally at three separate time points: twenty-four hours post ACL rupture, one month following ACLR surgery and ten months following ACLR surgery. Brain wave activity was observed using a CGX EEG head set at each time point. Congruently, the athlete participated in a contemporary evidence-based ACL rehabilitation protocol throughout all time points as outlined in Appendix A: Rehabilitation Summary Outline.11,12 The expected limitations in functional mobility early in the rehabilitation progression restricted some physical testing during the early testing dates; however, normal rehabilitative functional parameters were implemented to assure return to play status over time.
Explanation of Outcome Measures
Brain activity-dependent variables were measured using the CGX EEG head set. Cognitive workload metric, cognitive attention metric, sensorimotor rhythm (SMR) asymmetry, galvanic skin response (GSR), and three-dimensional cortex connectivity were derived to account for the central nervous system and brain precision and efficiency.13 Cognitive workload metric represents the depletion of mental resources of the brain due to higher levels of cognitive or sensory demands placed on the brain.14 GSR measures sympathetic conductance and electrical activity via a skin electrode which offers insight into the emotional state, or arousal, of a subject under specific experimental conditions. CNS arousal is typically directed by the thalamus with an emphasis on processing sensory inputs from the vision and hearing centers of the brain.13 Brain attention refers to a person’s ability to attain, remember, understand, and apply information.13 Heart rate (HR), heart rate variability (HRV), respiration rate (RR), peripheral body temperature, and superficial trapezius EMG activity were measured. Increases in HR, RR, and trapezius EMG activity are associated with elevated levels of central nervous system stress or increased GSR, whereas, HRV decreases with higher levels of stress.15
Neurocognitive and physical psychometric measures consisted of the Stroop test,16,17 visual reaction,17,18 a reaction time test,17 musculoskeletal range of motion, strength, a functional movement assessment consisting of balance-mobility-gait tasks,11,12 and force plate outputs for jump and landing mechanics on a ForceDecks (VALD Performance, Charlotte NC) force plates.19,20 The Stroop test is a neuropsychological test used to assess the speed or ability to process cognitive interference, inhibition, and selective attention capacity relative to simultaneous stimuli.16,17 The visual reaction was measured using the Dynavision D2, (Cincinnati, OH) A*-Proactive assessment which requires the individual to react and strike lights as they illuminate as quick as possible when presented in a random, computer-selected algorithm. In a similar fashion, the decision reaction time tests measured the response speed of the left and right hand and left-to-right brain activity precision prompted by visual stimuli.18 The functional movement and force plate assessments were used to measure bilateral functional asymmetries and relative ground reaction force upon walking gait, balance, landing, and jumping activities. Specifically, the force plate offers insight to force acceleration/decelerations, reactions, and bilateral imbalances. Overall, each measure provides an initial marker for longitudinal progressions that offer insight to assist in monitoring changes in functional performance and left-to-right symmetries of the limbs and brain function. The data was analyzed using CLR Neurosthenics, Inc neuroanalytics provided by Intheon Labs (San Deigo, CA).
The initial qEEG assessment was performed twenty-four hours following injury while the athlete was being monitored and treated to manage the acute stage swelling and pain. Non-weight bearing/crutch ambulation, ice, compression, elevation, and linear mobility exercises were initiated to promote improvement in passive and active ROM with reductions in pain and swelling. The state of the acute injury limited the ability to perform extensive functional and physical assessments. Immediately following injury, the reduction in the motor neuron network demonstrates a potentially altered signal from the peripheral receptors.21 Therefore, the initial tests used to monitor neurophysiological status using qEEG were the physiological regulatory measures, the Stroop test, decision reaction time, attention, workload, and SMR metrics. There was no pre-injury baseline.
One month post-surgical reconstruction, the athlete was progressing through a best-practice rehabilitation program previously reported to have sustainable, valid, and reliable techniques ideal for athletic populations and return-to-play progressions.11 Throughout the rehabilitation protocol the athlete progressed with improvements noted in pain, swelling, ROM, function, and functional joint stability.
Outcomes
Heart rate, heart rate variability, respiration, and brain attention levels yielded high-stress
Determinants in the acute stages of injury were associated with brain dysregulation, hemispheric asymmetries (Tables 2 and 3) and musculoskeletal functional asymmetries. Traditional objective measures of knee joint laxity, range of motion, swelling, strength, and patient-reported outcomes were collected throughout the rehabilitation progression and are outlined in Tables 4, 5, and 6. Central nervous system, brain performance, and musculoskeletal functional biomarkers were monitored longitudinally at three separate time points following anterior cruciate injury: twenty-four hours post ACL rupture, one month, and nine months which was the time period for her return to competition following ACLR surgery. Brain and musculoskeletal dysfunction longitudinally reveal a neurophysiological acute compensation and recovering accommodation from time points one to three. In addition, biological responses to stress, neuroanalytics of brain workload, arousal, and attention all advanced to more commonly regulated function during the progression of the rehabilitation. The neurophysiological development following acute ACL rupture demonstrates significant dysfunction in asymmetrical functions both neurocognitively and physiologically.
DISCUSSION
The original intent of this case was to investigate the innovative use of qEEG and neurophysiological metrics to monitor the longitudinal and simultaneous change in brain and central nervous systems activity while measuring musculoskeletal function during an anterior cruciate ligament repair rehabilitation. The data captured initial brain dysregulation and signs of accommodation that were simultaneously associated with muscular dysfunction over the nine-month period. Other authors have monitored only qEEG activity during isolated positional (kinesthetic) activity or only active ROM to demonstrate changes in brain wave activity.3,4,9 This is the first report to examine the use of the CGX EEG headset to perform a qEEG to evaluate and demonstrate simultaneous changes in both the central nervous system and the peripheral musculoskeletal system throughout an ACLR rehabilitation sequence.
The initial data regarding hemispheric asymmetries (Table 3) indicates the athlete’s brain was in a dysregulated state at initial injury and one-month following injury, but not at nine months following injury. A difference in brain activity, as little as 10-15% between each hemisphere, appears to create a dysregulated state.10 The initially slower right-handed reaction times indicate potential left hemisphere disruption as a result of having to accommodate for the injury to the right knee.21 In comparison, the left-hand reaction time was not clinically significant between time points one and two; however, it did demonstrate noted clinical improvements at nine months. The right hand was vastly different for all time points revealing a potential lag or inhibition in motor planning.21 The Dynavision D2 assessments from one month and nine months post-surgery revealed no significant change in performance which may indicate visual reaction time and accommodation following injury occur within the first month of rehabilitation and recovery. Recent literature has revealed both accuracy and reaction times to be impaired following ACLR.21 Conversely, this case data revealed slower reaction times following injury but no lag in accuracy. The results of the Stroop test remained constant throughout the rehabilitation program while the speed of responses significantly improved between each time point (Table 2).
The dysregulation noted in this case is likely multifactorial and associated with a compromised motor planning sequence. Recent literature has identified a potential inhibition response due to an increased strain on brain connectivity commonly associated with ACLR patients.21 The qEEG along with the neurophysiological measurements provides a clearer understanding of how the neural mapping of the dysregulated brain state and neurocognitive function are impacted during ACLR recovery. Bruns et al., reported hemispheric decoupling can be altered based on a task.10 Thought to be the result of lower corticospinal excitability in ACLR patients, lags in neurocognitive reaction time following injury appears to create a widespread cortical inhibition contributing to poor neural connectivity and peripheral motor response time. Reaction time and motor response play a vital role in athletic participation; thus, understanding the contributing factors of the CNS during ACLR recovery is critical to return to play progressions.
In the current case, attention and workload metrics were elevated beyond normal levels immediately following injury and progressively decreased over time regardless of physical or visual tasks. The workload metric was notably different initially, likely due to the CNS accommodating following the injury.4,21,22 As a result, the initial workload values were elevated and trended higher with physical demands of jump landing, but not for the simple functional movements. The immediate elevation in the attention and workload metrics is consistent with previous reports of injury decreasing brain connectivity and efficiency following injury.22 The increased workload and attention are accommodation mechanics thought to stimulate alternative neuronal activity directed on returning peripheral limb function.21 In addition, when an individual is under high cognitive workload and the cognitive workload approaches the individual’s cognitive capacity, human errors often occur.14 Overall, hemisphere regulation was regained over an 8 month time frame, as indicated by a two-percent differential in brain hemisphere symmetries (see Table 3). It is important to note that the asymmetries that occurred in the central nervous system and brain regions of interest were associated with similar accommodations and asymmetries in musculoskeletal function (See Tables 2 and 3).
Re-establishing normal function to the pre-injury level is the primary goal of rehabilitation after ACLR. The rehabilitation program used in the current case consisted of functional progressions normally seen in a traditional ACLR protocol. Return-to-play criteria rely on physical and clinical assessments related to range of motion, kinesthetic awareness, balance, and strength. The functional movement and force plate assessments used in the current study helped to guide the functional progressions necessary to advance movement patterns safely in preparation for return to sport. Throughout any rehabilitation process, left to right musculoskeletal asymmetries and functional capacities are used to evaluate exercise progressions. The current case reveals that monitoring the neurophysiological levels and brain electrophysiology activity in tandem with musculoskeletal function may be beneficial in maximizing return-to-play decisions post-injury or surgery.21,22
CONCLUSION
The objective of this paper was to demonstrate alterations in cortical brain activity after suffering an ACL injury and undergoing ACLR with the goal of returning to competition. The lack of cortical wave continuity, hemispheric asymmetries, and hemispheric decoupling throughout the phases of rehabilitation seem to support the need to monitor neurophysiological adaptations during rehabilitation to assure neural continuity between the peripheral and central nervous system.
The athlete advanced through the clinical pathway based on the traditional somatic metrics. While this case report demonstrates the novel use of qEEG to monitor brain neurophysiological somatic “readiness” for return to play/activity, more research is needed in order to support a causal relationship between these physical and neurological variables. Future studies with a larger cohort will be necessary to validate these conclusions. However, based on the data from this initial case study qEEG appears to have the potential to aid rehabilitation progressions by offering clinicians a collective insight into the neurological and physiological functional capacity of an ACLR patient.