INTRODUCTION
Shoulder pain is one of the most common musculoskeletal complaints, affecting up to 26% of adults at any given time and ranking third among musculoskeletal disorders seen in primary care settings.1–3 It is also associated with substantial functional limitations, work absenteeism, and increased healthcare utilization.1–3 Rotator cuff–related pathology is frequently implicated, and many individuals present with strength deficits despite the absence of full-thickness tendon tears or overt structural abnormalities on imaging.4–8
The mechanisms underlying rotator cuff weakness in the absence of tendon rupture are not fully understood. Pain-related neuromuscular inhibition can reduce voluntary activation, leading to reductions in force output, although the underlying source of pain may include structural pathology such as tendinopathy, partial tearing, or joint degeneration.9,10 Chronic disuse or denervation may result in reduced muscle cross-sectional area (CSA) while age-related fatty infiltration can impair contractile quality independent of muscle size.11–16 These processes may overlap; pain may initiate protective disuse, which could contribute to structural changes. Demographic factors such as age and body mass also influence baseline CSA, further complicating interpretation.11
Prior studies examining rotator cuff CSA and morphology have primarily focused on individuals with full-thickness tears, where reductions in muscle size are consistently observed.17–19 However, whether similar changes occur in patients with shoulder pain and structurally intact tendons remains unclear.8,12 Additionally, the association between rotator cuff muscle CSA and clinically observed strength and endurance deficits is unexplored in this group.8,18,20 Clarifying these relationships is critical for determining whether weakness reflects neuromuscular inhibition or true structural atrophy, which can be quantified through measurement of rotator cuff CSA.9,10,14,15 Neuromuscular inhibition may resolve rapidly with pain management and motor retraining, whereas atrophy requires longer rehabilitation targeting hypertrophy and load progression.21 Magnetic resonance imaging (MRI) has historically served as the reference standard for evaluating rotator cuff muscle morphology due to its high spatial resolution. However, ultrasound has emerged as a reliable and accessible alternative for measuring rotator cuff CSA, with prior work demonstrating strong agreement with MRI.22
Other factors may also influence CSA and performance. Dominant shoulders are typically exposed to greater habitual loading, potentially yielding greater muscular CSA regardless of pathology.23 Age, body size, and symptom chronicity may also influence morphology. Clarifying these relationships in both symptomatic and asymptomatic individuals may help distinguish normal adaptation from changes driven by rotator cuff-related shoulder pain.
The purpose of this study was to compare the SS and IS CSA and SS tendon thickness between painful and asymptomatic shoulders. Secondary aims were to examine the effects of limb dominance and to explore relationships between CSA, strength, and endurance. It was hypothesized that individuals with shoulder pain would demonstrate reduced CSA, strength, and endurance compared to controls, but that tendon thickness would not differ between groups. It was also hypothesized that non-painful dominant shoulders would exhibit greater CSA than non-dominant shoulders, without differences in strength or endurance. Finally, the relationship between CSA and symptom chronicity, age, or weight was explored.
METHODS
Participants
Two groups were recruited for this study: individuals with shoulder pain and asymptomatic controls. The shoulder pain group consisted of right-hand dominant individuals recruited from an ambulatory orthopedic practice for evaluation of unilateral right shoulder pain of various etiologies, representing a clinically heterogeneous population. Since limb dominance rather than absolute side was the variable of interest, comparisons were conducted using dominant versus non-dominant shoulder terminology, even though all participants reported right-hand dominance.
All symptomatic participants underwent MRI evaluation of the affected shoulder prior to enrollment. MRI reports were reviewed to confirm the absence of full-thickness rotator cuff tears. Individuals with MRI-confirmed full-thickness tears were excluded. Other structural findings (e.g., tendinosis, partial-thickness tears, labral pathology, or degenerative joint changes) were not exclusionary, as the study aimed to evaluate muscle morphology and function in a clinically representative population with intact rotator cuff tendons (Appendix A).
This study drew participants from a larger project originally powered for correlation analyses between CSA and strength. The case-matched control group consisted of a convenience sample of asymptomatic participants recruited through an internal email advertisement through the hospital intranet, and the patient population without shoulder pain from the ambulatory orthopedic clinic, where this study was conducted. Case-matched controls were matched by gender, age (similar decade of life), height (± 3 inches [7.62cm]), and weight (±10 lbs. [5.54Kg]). (Table 1)
Participants in both groups were eligible if they were ≥18 years of age and right-hand dominant. Exclusion criteria for all participants included prior shoulder surgery, shoulder pain, or MRI evidence of a full-thickness rotator cuff tear. Case-matched control participants were additionally required to have no shoulder pain within the prior three months.
After providing informed consent, each participant completed a brief questionnaire capturing demographic information and patient-reported outcomes. Ultrasound (US) testing was then scheduled. Each participant was assigned a unique participant identification number generated using a random number generator, which blinded the data analyst (DJK) to group allocation. However, blinding during testing was not feasible, as the primary investigator (DJK) was responsible for participant recruitment and data collection. This study was approved by the Institutional Review Board of Maine Medical Center, and all participants provided written informed consent before enrollment.
Height and weight were recorded at the time of ultrasound examination, and body mass index (BMI) was subsequently calculated. For participants in the shoulder pain group, pain intensity was assessed using both a numeric rating scale (NRS) and a visual analog scale (VAS).
All participants completed the modified Constant Score, the Penn Shoulder Score, and a single-assessment numeric evaluation (SANE), which asked, “What percentage of normal does your shoulder feel today?”24–27 The modified Constant Score (MCID = 10.4, MDC = 23) is a widely used composite measure that allocates 35 points to subjective assessments of pain and activities of daily living, and 65 points to objective measures.24,28 The Penn Shoulder Score (MCID = 11.4, MDC = ±12.1) is a 100-point patient-reported outcome measure comprising three subscales: pain, activities of daily living, and function. The SANE score (MCID = 15%) provides a single-item global rating of shoulder function and has demonstrated concurrent validity with the American Shoulder and Elbow Surgeons (ASES) score.25,29,30
Ultrasound testing
Ultrasound imaging of the SS and IS muscles was performed following a previously established protocol with demonstrated concurrent validity and intra-rater reliability (ICC 0.93–0.97) for measuring CSA. All ultrasound imaging was performed by the same primary investigator (DJK), who completed specialized coursework in musculoskeletal ultrasound under the supervision of an experienced co-investigator (CT) and had previously demonstrated excellent intra-rater reliability using this protocol.22
Participants were seated with their arm in neutral rotation at their side (0° abduction and elevation). For CSA measurements, panoramic ultrasound imaging was used. Anatomical landmarks, including the coracoid process and the medial and lateral borders of the scapular spine, were palpated and marked. A reference line (OBRL-1) was drawn from the lateral border of the scapular spine to the coracoid process. The midpoint between the medial and lateral scapular borders was then identified to denote the location of maximal CSA. A second line (OBRL-2) was drawn parallel and inferior to OBRL-1, extending to the inferior border of the scapula. The transducer was placed parallel to OBRL-2 and adjusted until the cortical bone appeared hyperechoic and sharply defined, indicating perpendicular alignment to the underlying tissue. CSA images of the SS and IS were captured and analyzed using the ultrasound system’s pencil-tracing function.22
For supraspinatus tendon imaging, participants were positioned in the modified Crass position (hand placed on the ipsilateral posterior iliac crest, with the elbow directed posteriorly). The bicipital groove was identified in the short axis view, and the transducer was then moved laterally to visualize the SS tendon. A transverse image was captured with the intra-articular portion of the biceps tendon in view. Tendon thickness was measured using the lateral edge of the biceps tendon as a reference point. Caliper measurements were taken at 5 mm and 10 mm lateral to this reference, and the average of these two values was used for analysis (Figure 1). This method has been previously described and shown to have high reliability (ICC = 0.93–0.97; SEM = 0.2–0.3 mm; MDC = 0.6 mm).31
Strength and endurance
Isometric strength and muscular endurance were assessed using handheld dynamometry and the Posterior Shoulder Endurance Test (PSET), respectively.32 All strength measurements were performed using a MicroFet 2 handheld dynamometer (HHD) and a standardized “make test” protocol.33
Strength of the external rotators was assessed with participants standing, shoulder in neutral rotation, and the elbow flexed to 90° with a towel roll placed in the axilla to maintain neutral abduction. Participants stood with their feet shoulder-width apart to provide a stable base of support while resisting the dynamometer applied to the posterior aspect of the wrist. (Figure 2A). Strength during the full can test was assessed with participants standing with the shoulder elevated to 90° in the scapular plane and the thumb oriented upward. The dynamometer was placed at the radial styloid of the distal forearm (Figure 3B). Participants were instructed to push maximally into the device for three seconds while verbal encouragement was provided. Participants with shoulder pain were instructed to exert maximal effort up to their tolerable pain level. Pain experienced during testing was recorded using a 0–10 numeric rating scale, and the maximum pain reported during each strength test was used for analysis. Three trials were performed for each movement, and the average value was used for analysis. Pain intensity was rated on a 10-point Likert scale following each isometric contraction.
Muscular endurance was assessed using the PSET as described by Moore et al. Participants lay prone on a treatment table with the test arm hanging off the edge (Figure 3).32 A dumbbell equivalent to 2% of the participant’s body weight was used for testing. Participants raised their arm in horizontal abduction with external rotation, with the thumb pointing toward the ceiling, to 90° at a cadence of 30 beats per minute, guided by a metronome. The test was terminated upon the first instance of fatigue, defined as an inability to reach 90°, compensatory trunk movement, or verbal report of fatigue. A pitch counter was used to record the number of successful repetitions. Participants rated their shoulder pain on a 10-point Likert scale following completion of the test for each arm.
Data Analysis
All statistical analyses were conducted using SPSS version 25. An alpha level of 0.05 was set a priori for all tests. Independent t-tests were performed to ensure there were no differences between the case-controls and symptomatic groups at baseline for group matching on age, height, and weight.
To evaluate the primary and secondary aims, a 2 (group: shoulder pain vs control) × 2 (arm: dominant vs non-dominant) mixed-model analysis of variance (ANOVA) was performed to assess differences in SS and IS CSA, SS tendon thickness, isometric strength (external rotation and full can), and posterior shoulder endurance. Post hoc comparisons were conducted using Tukey’s HSD in the presence of significant main or interaction effects.
To explore potential covariate influences on CSA, a follow-up analysis of covariance (ANCOVA) was performed with height, weight, and symptom chronicity as covariates. If group differences in strength or endurance were observed, additional ANCOVA models were run using pain reported during testing as a covariate to evaluate its contribution to performance outcomes.
Finally, a three-step hierarchical multiple regression was performed to examine whether SS/IS CSA and pain during testing predicted isometric strength (external rotation and full can). Simple linear regression was also conducted to assess the relationship between SS CSA and tendon thickness.
RESULTS
Participants
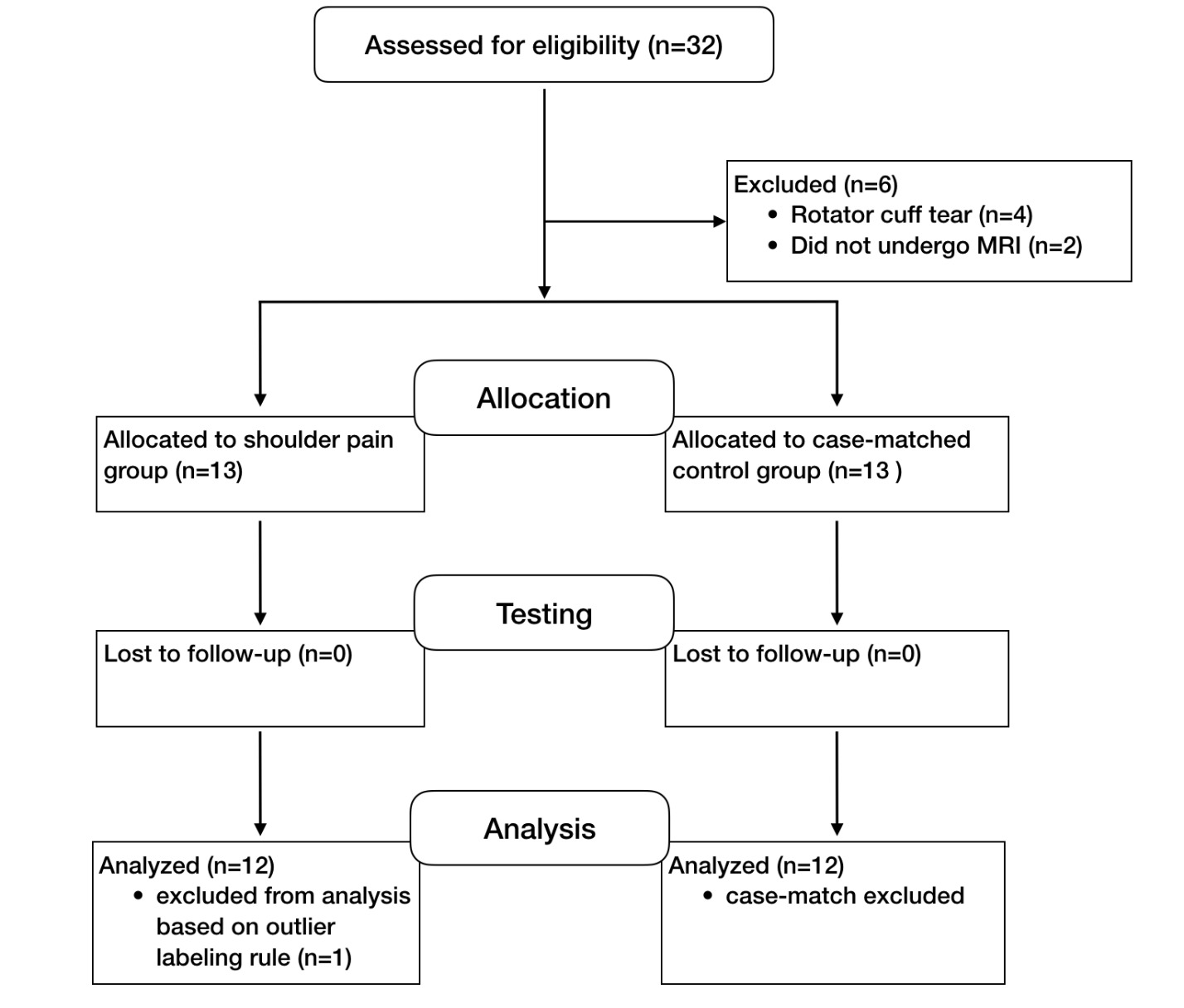
A total of 32 participants were screened for eligibility. Four individuals were excluded due to full-thickness rotator cuff tears. One participant in the shoulder pain group was excluded as an outlier using Tukey’s resistant outlier labeling rule (values exceeding 1.5 times the interquartile range from the upper or lower quartile), and their matched control was also removed to preserve case-matching integrity.34 This resulted in a final sample of 24 participants (48 shoulders) included in the analysis (Figure 4).
The shoulder pain group included 12 right-hand dominant individuals (5 female; mean age = 43 ± 12.5 years), all with unilateral right shoulder pain. The control group consisted of 12 case-matched individuals (5 female; mean age = 45 ± 11.2 years). No significant differences existed between groups in age, height, or BMI (p > 0.05; Table 1).
Participants in the shoulder pain group reported an average symptom duration of 8.6 months. Pain was most commonly provoked by strenuous activity and sleeping. MRI findings in the symptomatic cohort demonstrated heterogeneous structural abnormalities typical of rotator cuff–related shoulder pain, including tendinosis, partial-thickness tears, acromioclavicular joint arthrosis, labral pathology, and adhesive capsulitis (Appendix A). No participant included in the final analysis demonstrated a full-thickness rotator cuff tear. This group reported greater disability than controls across all outcome measures, and overall irritability was categorized as moderate based on a treatment-based classification model.35 Three participants in the shoulder pain group did not complete the PSET: two due to limited range of motion, and one due to pain. Therefore, PSET analyses for the symptomatic group were based on n = 9. All other outcome measures were completed by all included participants.
Cross-Sectional Area and Tendon Thickness
No significant main effects or interactions were observed for SS or IS CSA (all p > 0.19; Table 2). Similarly, no differences in SS tendon thickness were found between groups or sides (p > 0.86; Table 2). Linear regression analyses also demonstrated no significant association between SS CSA and tendon thickness (p > 0.05).
Strength and Endurance
Results of the strength and endurance analyses are summarized in Tables 3 and 4.
Isometric External Rotation (ER) Strength
There was a main effect of limb dominance (p = 0.006). Within the shoulder pain group, the painful dominant shoulder was significantly weaker than the contralateral side (mean difference = 1.7kg, p = 0.003) and was 3.3kg weaker than the dominant shoulder in controls, though this did not reach a statistically significant difference (p = 0.058). When pain during testing was entered as a covariate, it explained 36% of the variance and eliminated the dominance effect, suggesting that pain largely accounted for the observed weakness. (Figure 5)
Full Can Strength
There was a main effect of dominance (p = 0.013) and a group × dominance interaction (p = 0.024). Painful dominant shoulders were significantly weaker than both the contralateral side (p = 0.003) and the dominant side in controls (p = 0.035). No strength asymmetry was observed in controls (p = 0.329). When pain during testing was entered as a covariate, the interaction was no longer significant, and pain itself did not independently contribute to the model. (Figure 6)
Posterior Shoulder Endurance (PSET)
A main effect of dominance (p = 0.029) and a group × dominance interaction (p = 0.032) were found. Painful dominant shoulders completed ~13 fewer repetitions than the contralateral side and ~17 fewer than the dominant side in controls (both p < 0.05). No differences were found for the non-dominant side (p = 0.50). When pain was included as a covariate, it significantly influenced endurance outcomes (p = 0.038), but group differences persisted, suggesting additional contributors beyond pain. (Figure 7)
Regression Analyses
A hierarchical multiple regression analysis was conducted to determine whether pain during testing and the CSA of the SS and IS muscles predicted isometric force production (full can and ER strength) and PSET. The regression models examined the relationship between rotator cuff CSA and strength across the full sample. Table 4 summarizes the results of each model.
For full can strength, SS CSA explained 49% of the variance in full can strength (p < 0.001). Adding pain during testing significantly improved the model, increasing the explained variance to 56% (ΔR² = .073, p = 0.008). Both SS CSA (β = .74) and pain (β = –.27) were significant predictors. Adding IS CSA did not significantly improve the model when entered in the final step (ΔR² = .017, p = 0.193).
For ER strength, IS CSA accounted for 33% of the variance in ER strength (p < 0.001) and remained the only significant predictor in subsequent steps. Pain (ΔR² = .024, p = 0.204) and SS CSA (ΔR² = .041, p = 0.093) did not significantly improve model fit.
For PSET endurance, pain was the only significant predictor of endurance performance, explaining 11% of the variance (β = –.33, p = 0.023). Neither SS nor IS CSA significantly contributed to the model when entered in later steps (both p > 0.14).
In summary, SS CSA predicted full can strength, IS CSA predicted ER strength, and pain during testing predicted endurance. Strength models were modestly improved by including pain, while endurance performance was explained only by pain ratings.
DISCUSSION
In individuals with unilateral shoulder pain and MRI-confirmed intact rotator cuff tendons, SS and IS CSA did not differ from asymptomatic shoulders, despite heterogeneous structural findings typical of rotator cuff–related shoulder pain. These findings included tendinosis, partial-thickness tears, labral pathology, and degenerative joint changes, which may contribute to pain and neuromuscular inhibition even when full-thickness tendon rupture is absent. Strength and endurance deficits were observed, suggesting that functional impairments in this population are not necessarily accompanied by measurable reductions in muscle size.
Although SS and IS CSA were predictive of full can and external rotation isometric strength, respectively, the strength of these relationships was moderated by the influence of pain. In pain-free populations, the association between rotator cuff CSA and strength is strong. For example, Gerber et al. performed suprascapular nerve blocks in healthy individuals and found that abduction and external rotation strength were substantially impaired (>70%).9 Similar patterns have been observed in overhead athletes with selective infraspinatus atrophy.20 The weaker association observed in this cohort suggests that pain-mediated inhibition disrupted the typical relationship between CSA and strength, indicating that functional weakness in the presence of intact tendons is more likely explained by pain-related inhibition than by atrophy.
Pain can impair muscle performance through several mechanisms, most notably neuromuscular inhibition that limits voluntary activation.10,13,21 Disuse and neuropathic changes may also contribute, and in chronic states, these mechanisms often lead to measurable atrophy.13–16 In this cohort, however, pain contributed to strength deficits but was not associated with reduced CSA, suggesting that moderate, chronic pain in the absence of tendon rupture may inhibit force production but without measurable reductions in the cross-sectional area of the SS or IS.
The inhibitory effects of pain on muscle performance are well documented through the phenomenon of arthrogenic muscle inhibition, in which joint pain or pathology reflexively limits motor unit activation.21 Stackhouse et al. demonstrated that experimentally induced subacromial pain reduced external rotation strength and voluntary activation, but only when pain intensity exceeded a moderate-to-severe threshold (~7/10).10 In the present study, peak pain during testing was lower (≤5/10), which may explain why deficits in force production were observed without measurable reductions in CSA. These findings suggest that moderate pain is sufficient to inhibit activation but not to reduce CSA when tendons remain intact; however, it cannot be ruled out that more severe or prolonged pain states may yield different results. The highest reported pain during external rotation testing was 5/10. Yet, adjusting for pain eliminated observed between-group differences in strength, with pain accounting for over one-third of the variance. Together, these findings support the interpretation that pain-mediated inhibition, rather than structural atrophy, was the primary contributor to weakness in individuals with intact rotator cuff tendons. Consequently, these findings suggest that pain reduction and restoration of neuromuscular control may be important early considerations in rehabilitation, with progressive strengthening introduced as pain and muscle activation capacity improve.36,37
Despite an average pain duration of 8.6 months, no between-group differences in supraspinatus or infraspinatus CSA were observed. Multiple mechanisms may account for preserved CSA despite the chronicity of pain. First, participants in the present study likely maintained partial daily use of the painful shoulder, which may have provided enough mechanical loading to preserve muscle size. Second, while pain can inhibit voluntary activation and reduce strength, the intensity and irritability levels in this cohort may not have been high enough to drive substantial disuse atrophy. Finally, the intact tendon structure in all participants preserved the potential for normal force transmission, increasing the likelihood that the observed weakness reflected pain-related neuromuscular inhibition. Future studies should investigate whether higher irritability pain states, activity restrictions, or post-surgical immobilization in non-tear populations result in detectable reductions in rotator cuff CSA.
Endurance performance, as assessed by the PSET, was not significantly associated with SS or IS CSA. While pain negatively influenced PSET outcomes, differences between painful and asymptomatic shoulders persisted after adjusting for pain, indicating that additional factors contribute to the observed endurance deficits. The PSET reflects coordinated activity across the posterior shoulder complex, including the infraspinatus, posterior deltoid, and scapular stabilizing musculature. Electromyographic investigations have demonstrated substantial activation of both rotator cuff and scapular muscles during the test, indicating that performance reflects multi-muscle endurance rather than the function of a single muscle group.38 Therefore, reduced endurance in the symptomatic cohort may reflect impairments in posterior shoulder neuromuscular performance rather than isolated rotator cuff muscle size.38 This interpretation is consistent with Sozlu et al., who demonstrated that scapular muscle endurance is reduced in patients with rotator cuff–related shoulder pain,39 and aligns with broader mechanistic models of recovery in rotator cuff tendinopathy that emphasize neuromuscular adaptations as a pathway for improvement.6,37 These findings suggest that rehabilitation may need to address scapular muscle performance and neuromuscular coordination alongside pain reduction, rather than focusing solely on rotator cuff strengthening. Notably, three participants were unable to perform any repetitions due to range of motion limitations or pain, reducing the analyzable sample for this outcome to n = 9. This may have led to an underestimation of endurance capacity in the painful group and limits confidence in the between-group comparisons for this measure.
This study has several limitations. First, participants in the painful group were not specifically screened to differentiate glenohumeral or subacromial pathology from other potential contributors, such as cervical radiculopathy or scapular dysfunction. Although all were referred from a specialty orthopedic practice and most had MRI-confirmed glenohumeral findings, alternative pain sources cannot be fully excluded. Second, participants generally exhibited moderate tissue irritability and retained functional use of the shoulder, which may have limited the likelihood of detecting CSA differences. Higher irritability or greater disability may be necessary to induce measurable atrophy. Third, participants were matched by age decade to control for age-related differences in muscle morphology and strength broadly. However, upper extremity activity level was not formally quantified, and differences in habitual loading, sport participation, or occupational demands may have influenced strength or endurance outcomes. Additionally, all participants in the shoulder pain group presented with right-sided symptoms, which may limit the generalizability of these findings to individuals with left-sided shoulder pain. Fourth, this investigation was conducted as part of a larger project, with the original power analysis based on correlation rather than between-group comparisons. Therefore, group analyses were exploratory and may be underpowered to detect small-to-moderate effects. The exclusion of seven participants due to full-thickness tears, outliers, or loss to follow-up further reduced the sample size, which increases the risk of type II error in the regression analyses. Finally, strength measurements were obtained using hand-held dynamometry, which quantifies applied force rather than joint torque. Because individual differences in limb length were not measured, potential differences in moment arm between participants could influence torque generation at the shoulder and may limit the precision of between-subject strength comparisons.
Future studies should investigate whether more severe or acute shoulder pain, particularly in patients with higher tissue irritability, produces measurable reductions in rotator cuff CSA. Populations in the early post-injury or post-surgical phases may be especially informative, as pain and protective movement patterns are often most pronounced. Longitudinal designs that track CSA, tendon morphology, and muscle performance from symptom onset through recovery would help clarify the temporal relationship between pain, disuse, and atrophy. In addition, adequately powered studies that account for potential confounders such as limb dominance, activity level, and comorbidities are needed to better define how CSA relates to functional strength outcomes. Future investigations incorporating direct measures of scapular and rotator cuff muscle activation may also help clarify the contribution of posterior shoulder musculature to endurance performance.
CONCLUSION
In individuals with moderate shoulder pain, SS and IS CSA did not differ between painful and asymptomatic shoulders or between groups, despite significant deficits in strength and endurance. Although CSA was predictive of isometric strength, these deficits were not explained by atrophy. Instead, the findings suggest that weakness in this population is more likely driven by pain-mediated inhibition or other neuromuscular mechanisms than by structural changes, which may have implications for rehabilitation strategies.
Conflicts of interest
The authors declare that they have no conflicts of interest related to this study.
Funding
This research was partially funded by a mentored research grant from the Maine Medical Center Research Institute.
Acknowledgements
We would like to thank Dr. William Dexter and Dr. Wendy Craig of Maine Medical Center for their expertise and assistance with this research, and Margaret Dolan for her eternal support.
_panoramic_view_of_the_supraspinatus_muscle_for.png)
_external_rotation_assessed_with_the_elbow_flexed.png)
._(a)_starting_position_with_the_arm_hanging_off_t.png)
.png)
.png)
.png)
_panoramic_view_of_the_supraspinatus_muscle_for.png)
_external_rotation_assessed_with_the_elbow_flexed.png)
._(a)_starting_position_with_the_arm_hanging_off_t.png)
.png)
.png)
.png)