INTRODUCTION
Injury to the anterior cruciate ligament (ACL) requires a long road to recovery to return patients to their desired activity, often including ACL reconstruction (ACLR) and a strenuous post-operative rehabilitation.1 Rehabilitation specialists, such as physical therapists, work closely with the injured patient, referring provider, and other members of the care team to ideally create an evidence-based, but personalized, plan of care. This is often guided by a post-operative rehabilitation protocol commonly provided by the treating orthopedic surgeon. Though there are no licensing requirements that require following the prescribed rehabilitation protocol and state laws may influence the scope of practice, rehabilitation specialists may be liable if adverse events occur if practicing outside of the prescribed protocol.2,3 Though most post-operative protocols allow flexibility for the rehabilitation specialist to individualize the rehabilitation to the needs of the patient, variability regarding timeline for patient progression, permitted interventions, and return-to-activity criteria may influence patient treatment and outcomes.
Patients following ACLR present with a large variability of rehabilitation needs, commonly driven by their post-operative goals, concomitant surgical procedures, and the recovery from injury/surgery. As such, post-operative therapeutic and exercise prescriptions should have appropriate flexibility to be individualized for each patient. This has been observed within publicly available rehabilitation protocols from academic institutions,4,5 with broad recommendations specific to the timing of strengthening exercise (i.e., quadricep sets, squats, etc.) and activities (bike, elliptical, running, etc.) being made. Patient outcomes post-ACLR are dependent on the improvements in strength, gait, and functional independence sought after in post-operative rehabilitation.6,7 Further, improvements of these outcomes are indicative of improvements in patient reported outcomes (PRO),8,9 emphasizing the importance of targeting post-operative prescriptions to these performance outcomes. However, the inconsistency of recommended assessments and clinical thresholds (i.e., limb symmetry thresholds) may provide challenges to rehabilitation specialists to guide rehabilitation progression or clearance decisions between patients and varying protocols.
The timing and frequency of rehabilitation visits has also been a topic of discussion within the ACLR literature. Functional deficits are commonly observed between five to nine-months post-ACLR.10 For instance, quadriceps strength six to nine months post-ACLR may only be 65 to75% of that observed in the contralateral limb.11,12 ACLR limb medial and lateral maximal hop for distance is also significantly reduced within the first year, by approximately 10-15%.13,14 As such, it is common for ACLR rehabilitation protocols to specifically prescribe rehabilitation interventions to target strength and functional deficits during these timepoints.3 However, majority of US insurance-backed physical therapy provides a number of visits that will be financially covered post-ACLR. A study of a total of 11,518 patients from a US health insurance database reported that half of post-ACLR physical therapy visits were completed within the first six weeks (1.4 months), and 90% of visits within the initial 16-weeks (3.6 months) following surgery.15 Secondly, a large focus of ACLR rehabilitation is the progression towards, and subsequent clearance for, return to sport (RTS). Timing of RTS has been highly reported on, with evidence showing the majority of patients six-months post ACLR, do not “pass” criteria of RTS protocols,16 and emerging evidence suggesting that early RTS may increase the probability of reinjury.17–19 Studies have demonstrated that following discharge from formal therapy programs, eight weeks of continued functional training can greatly improve measures of performance and patient reported outcomes.20,21 With this emerging evidence, questioning if post-ACLR rehabilitation protocols provide consistent timeline recommendations for the RTS progression appears important,16 to guide clearance decisions, as a survey of American orthopedic surgeons has reported inconsistencies with RTS timelines.22–24
Large variability amongst post-operative rehabilitation protocols specific to the time of exercise prescription has been reported,4,5 which may ultimately lead to large variability within patient outcomes. The current scoping review aims to characterize the variability in publicly accessible ACLR rehabilitation protocols from U.S. academic medical centers, with a focus on terminal phase timing and RTS criteria. The current review is not intended to provide the correct or optimal post-operative rehabilitation protocol or RTS testing battery. Rather, defining the variability amongst US rehabilitation protocols may draw attention to current rehabilitation protocols and be utilized to promote discussion and updates to existing protocols within surgical and rehabilitation teams.
METHODS
Protocol
This review follows guidance from the Joanna Briggs Institute Reviewer Manual and the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) to conduct the review and report results.
Eligibility criteria and Information sources
A scoping review was conducted to collect current, publicly accessible, post-operative ACLR rehabilitation protocols, in the English language, from institutional teaching hospitals identified by the Association of American Medical Colleges (AAMC). Protocols were obtained via institution and affiliated surgeon websites and were included in the review irrespective of graft type or meniscal involvement. Excluded protocols included those with concomitant ligamentous repair, ACL revision, and microfracture.
Search
On April 25, 2024, the AAMC Organizational Characteristics Database was utilized to identify the associated Medical School Details and Teaching Hospital Affiliates. Each teaching hospital affiliates’ website was then searched, by two independent authors, to identify publicly accessible protocols for ACLR rehabilitation. In the case where the hospital website directed users to a specific orthopedic surgeon’s site, that website was also searched for ACLR rehabilitation protocols. A second search was conducted on February 6, 2025, by a third independent author, to ensure that all publicly accessible protocols were identified. A secure cloud-based application was used for protocol organization. Found protocols were identified and saved under the AAMC institution, affiliated teaching hospital, specified surgeon, and specified ACLR surgery as applicable.
Selection of sources of evidence
Multiple ACLR protocols were collected for each affiliated teaching hospital/surgeon, if no standalone ACLR protocol existed (e.g. ACL BPTB autograft, ACL Hamstring autograft, ACL allograft, etc.). Protocols were categorized as 1) institution specific, 2) surgeon specific (independently created), or 3) third party (protocol outsourced from a named group other than current institution or physician group) i.e. the MOON ACLR protocol. In addition, protocols that were specific for ACLR plus meniscus repair were collected and categorized separately.
Data items and Data Charting Process
As this review is focused on the terminal phases of ACLR rehabilitation, specifically to the RTS process, the following variables were extracted from each protocol: date of protocol publication, specified graft type, inclusion of meniscal repair, stated date range of phases, criteria for advancement of phases, RTS criteria.
Synthesis of results
Specified graft type: each of the included protocols were grouped into which graft type the protocol was specifically intended for. These included non-specified autograft, hamstring autograft, bone patellar-tendon bone autograft, quadriceps autograft, allograft, or the graft site was not specified or was all inclusive for the graft sites a particular surgeon utilized. A percentage was then calculated for each of the graft type groups within the total number of included protocols.
Inclusion of meniscal involvement: each protocol was evaluated for including adjustments for meniscal involvement if they were not specifically labeled as a protocol specifically intended for ACL with meniscal repair, using a binary coding system where “1” was assigned to those that included adjustments and “0” for those that did not. A percentage was then calculated for those protocols that included adjustments for meniscal involvement.
Labeled and/or self-defined as “accelerated” protocol: each protocol was examined for labeling or statement of being an “accelerated” ACLR protocol.
Criteria for advancement of phases: each included protocol was evaluated for inclusion of criteria to advance to the terminal phase using a binary coding system where “1” was assigned to those protocols that did include criteria to advance and “0” assigned to those that did not. A percentage was then calculated for those that included criteria to advance.
Stated date range of terminal phase: for each included protocol, the stated start and end time for the terminal, or last, phase was identified. Mean and standard deviation were then calculated. If no end time was stated but instead was left open ended, those protocols were then grouped and its percentage within the total number of included protocols was calculated. Return-to-sport criteria: each included protocol was evaluated for inclusion of RTS criteria using a binary coding system where “1” was assigned to those that included criteria and “0” for those that did not. A percentage was calculated for those protocols that included RTS criteria within the total number of included protocols. What each protocol listed as the RTS criteria including any stated threshold that must be met, was then extracted. This is reported grouped by inclusion of strength, hopping, PRO, and measure of confidence and sub-grouped by their respective threshold value. A percentage was calculated for each specific criteria and its threshold value within the total number of included protocols that included RTS criteria.
RESULTS
Selection of sources of evidence
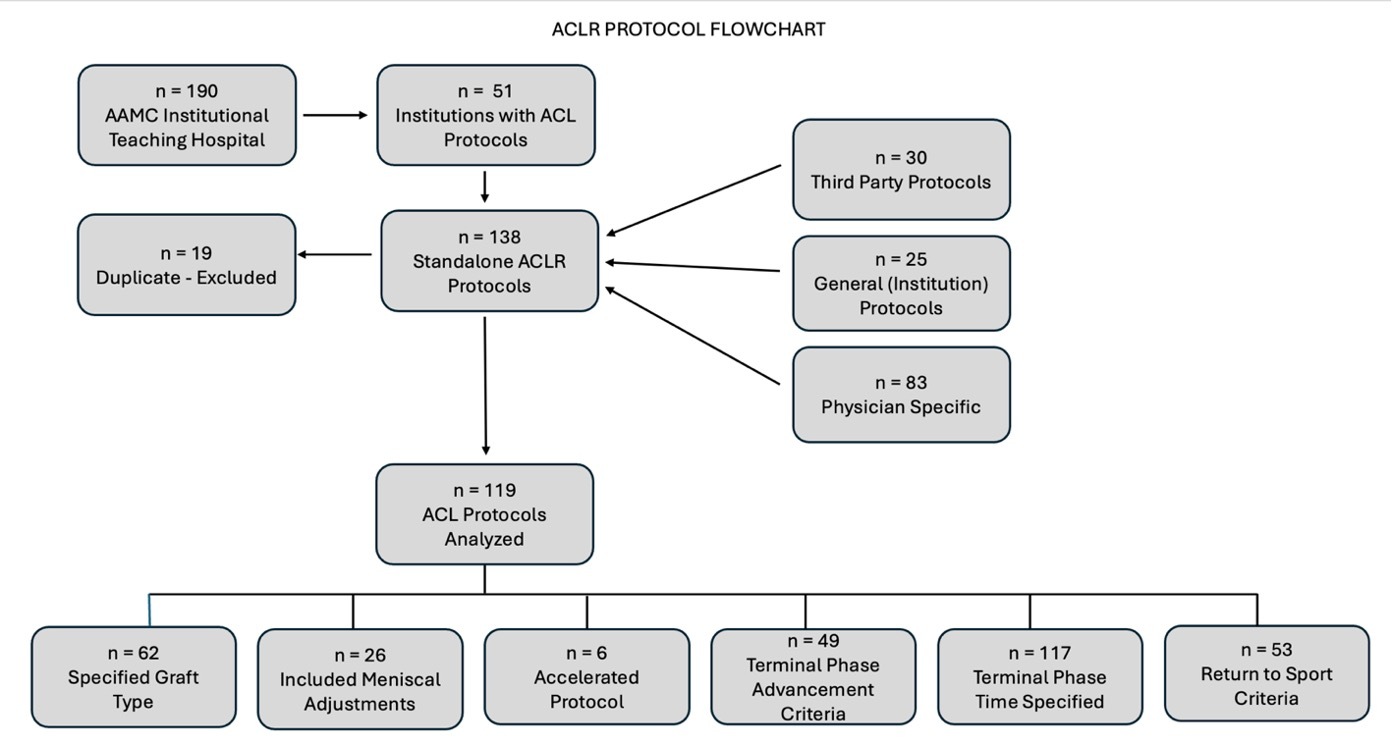
Out of 190 AAMC institutional teaching hospitals, 51 (26.8%) institutions had protocols publicly available, from which 138 standalone ACLR rehabilitation protocols were identified. Identified protocols were then categorized into one of three groups as follows: 30 third party protocols, 25 general (institution) protocols, and 83 physician specific protocols were noted. Of the 138 found protocols, 19 were removed for being a duplicate, with 119 standalone ACL protocols included for analysis (Figure 1). Of note, 46 protocols specific to ACLR plus meniscus were also identified during our search. Adjacent flowchart, protocol characteristics and results can be found within the supplementary material (Supplemental Figure A and Supplemental Table A and B).
Characteristics of sources of evidence
Of the 119 standalone ACL protocols included for analysis, 62 (52.1%) were graft specific, 26 (21.8%) included adjustments for meniscus involvement, 6 (5.0%) were accelerated, 49 (41.2%) included criteria for advancement to the terminal phase, 117 (98.3%) included a specified time for the final phase of rehabilitation, and 53 (44.5%) included RTS criteria (Table 1).
Results of individual sources of evidence (Table 2)
The average start of the terminal phase across the 117 protocols that included a specified time for the terminal phase was 5.42 ±1.79 months post-ACLR (range: 1.6-10.0 months, Figure 2). Of these, 65 (55.6%) provided either a time range or an endpoint for the completion of the terminal phase with an average of 7.38 ±2.73 months post-ACLR (range: 2.8-12.0 months). The remaining 52 (44.4%) protocols had an open-ended terminal phase, not specifying an endpoint for completion (e.g. Month 6+).
Of the 53 out of 119 ACLR protocols that included RTS criteria, 18 (34.0%) required various patient reported outcomes with 13 (24.5%) requiring a measure of patient confidence. Strength requirements were found in 79.2% (42 of 53) of protocols, with 34 of them requiring a particular strength threshold via limb symmetry index (LSI). Of the 42 protocols that recommended strength assessments prior to RTS, an LSI of 80% was included in 9 (21.4%), an LSI of 85% was included in one (2.4%), an LSI of 90% was included in 18 (42.9%), an LSI of 95% was included in two (4.8%), an LSI of 100% was included in 4 (9.5%), and 8 (19%) protocols recommended strength with no threshold provided (Figure 3). There was no prescribed methodology regarding how strength was to be assessed (i.e. handheld dynamometry, isokinetic dynamometry: mode, position, speed, etc.). Measure of hopping capabilities were found in 90.5% (48 of 52) of protocols, with 36 of them requiring a particular hopping threshold via LSI. Of the 48 protocols that recommended hopping assessments prior to RTS, an LSI of 80% was included in 3 (6.3%), an LSI of 85% was included in 12 (25%), an LSI of 90% was included in 12 (25%), an LSI of 95% was included in 5 (10.4%), an LSI of 100% was included in four (8.3%), and 12 (25%) protocols recommended hopping assessments with no threshold provided (Figure 3).
DISCUSSION
Large patient variability presents following ACLR, such as the time of RTS, time of concluding prescribed rehabilitation, and success of post-operative outcomes. As such, the purpose of the current review was to examine the publicly accessible physical therapy ACLR protocols for the conclusion of prescribed rehabilitation and clinical recommendations for returning to sport. The current review identified 184 ACLR rehabilitation protocols from 51 US academic hospitals. The average terminal phase of rehabilitation was from 5.42 ±1.79 months to 7.38 ±2.73 months post-ACLR. Of the 119 ACLR protocols identified, only 44% included RTS criteria, with the most common criteria being single leg hopping and lower extremity strength assessments.
The terminal phase of rehabilitation is often the last stage prior to the RTS progression. Some patients may have extended access to medical oversight from rehabilitation specialists from school- or athletic team-affiliated staff; however, for many patients, this phase marks the end of insurance backed rehabilitation from licensed personnel prior to self-prescribed treatment or out-of-pocket expenses. Without medical oversight, the end of the terminal phase may be met with the patient concluding rehabilitation, strengthening, or sport integration activities. Of the 119 protocols identified in the current review, 52 (44%) did not present an “end” timepoint but rather concluded their rehabilitation with timepoints that do not end (i.e. “12+” months). Of the 67 protocols that did specify an end timepoint of the terminal phase, the average end-time was 7.38 ±2.73 months post-ACLR (range: 2.8-12.0 months). Majority of ACLR patients conclude their prescribed post-operative physical therapy prior to 6 months post-ACLR.15,25 With current RTS timing recommendations by both orthopedic surgeons and physical therapists being closer to nine to 12 months post-ACLR,26 it presents the question of who patients turn to without medical guidance or oversight into achieving their rehabilitation goals. This is supported by research demonstrating 54% of patients complete self-prescribed exercise and rehabilitation at just over 6-months post-ACLR.25 Recent evidence has suggested a protective effect of time since surgery showing a greater risk of reinjury for patients returning to sport prior to eight- and nine-months post-surgery.
Large variability also presented in the criteria recommended for RTS. Of the 119 protocols reviewed within the current study, 55% of them provided no suggested RTS criteria. Though most rehabilitation specialists administer functional assessments that are not specifically included in the post-operative protocol,26 the inclusion of specified criteria may promote terminal phase assessments to identify functional deficits prior to the conclusion of insurance backed rehabilitation. It is also unlikely that a single RTS criteria would appropriately dictate the necessary sport demands for all patients following ACLR, given the large variability of activity, intensity, and patient factors that contribute to individualization of RTS progressions. However, the lack of criteria within prescribed post-operative protocols may leave ambiguous conclusions to the rehabilitation phase for patients. Large deficits of strength and functional performance exist at seven-months post-surgery,10,27 the average conclusion of the terminal phase that was found in the current review. Promoting the administration of functional testing around this time post-surgery may promote additional rehabilitation visits with professional oversight to better manage functional deficits and the RTS progression.
In looking at the specific criteria prescribed within the post-operative protocols; large variability exists within the assessments and the recommended thresholds provided. Of the 53 rehabilitation protocols that provided specific RTS criteria, only 34% required patient reported outcome measures and 25% of protocols required a measure of patient confidence. Single leg hopping requirements were the most common, found in 90.5% of protocols, and strength criteria was identified in 79.2% of protocols. This aligns with the criteria used to manage RTS decision making, as reported by Burgi et al in a 2019 scoping review,28 though they extracted RTS criteria from peer reviewed literature compared to the current review extracting criteria from publicly accessible post-operative protocols that patients receive. Only 25% of extracted protocols recommended any assessment of patient confidence within RTS testing. The psychological readiness to return-to-sport, often quantified through the ACL-RSI,29 has shown to be a predictor of patient outcomes, including the success of RTS and subsequent re-injury.30 Patient confidence post-ACLR has also shown to be independent of improvements in muscle strength or function,8,31,32 highlighting the need of including the assessment of patient confidence at the end of structured rehabilitation. Strength and single leg hopping were the most common reported RTS criteria, included in about 80% and 90% of protocols, respectively. As majority of patients post-ACLR demonstrate deficits within these assessments at the end of rehabilitation,33 the need for administering these assessments is supported. However, the utility of these assessments to predict successful outcomes of RTS with no subsequent reinjury has been challenged, with traditional assessments of knee extensor and flexor strength demonstrating no ability to predict reinjury.19,34–36 Strength and hopping testing may accurately assess the foundational components needed for RTS (i.e. power, peak torque, eccentric loading, linear deceleration, etc.), but further assessments indicative of sport-specific demands may be desired by rehabilitation teams and protocols to guide the RTS progression.35
Thresholds provided for objective strength and hopping assessments ranged from 80 to 100% limb symmetry index, with 90% LSI being the most common (Figure 3). Thresholds of strength and hopping provide a goal and milestone for both patient and rehabilitation provider, which may aid in rehabilitation compliance and patient motivation. The use of LSI measures following ACLR has also been presented with caution, as contralateral weakness has been shown to inflate LSI values.37,38 As such, thresholds using normative measures of strength, often relative to body mass (i.e. Nm/kg), have been published to predict outcomes post-ACLR.39,40 Though these measures present challenge regarding the interpretation and accessibility of equipment, no protocols within the current review provided recommendations of normative values strength as a target for patients following ACLR.
The current review is not without limitations. Rehabilitation protocols were extracted dependent on public availability, and institutions or hospital systems may not share rehabilitation protocols externally outside their medical network. The current review was also limited to public institutions identified by the AAMC, which could differ from post-operative protocols prescribed from private practices. However, the goal of the current review was to gather insight on the end-phases of prescribed rehabilitation via a nation-wide search. Through the 53 institutions, a similar number to other scoping review methodologies,4,5 and 119 protocols ranging 32 states, the current review can provide a general scope of post-operative protocols that are provided to patients following ACLR within the United States.
Conclusion
The results of this scoping review indicate that large variability exists within prescribed rehabilitation following ACL-reconstruction within US academic institutions. This variability ranges from the timing of the terminal phase of rehabilitation, recommendations of RTS criteria, and the specific criteria and thresholds used for RTS. Post-operative rehabilitation protocols can provide structure to the progression while also allowing room for individualization due to patient specific factors. Reviewing and updating post-operative protocols as a healthcare team may provide the greatest benefit to the patient and rehabilitation specialists.
Conflicts of interest
The authors have no conflict of interest to disclose.
._arrows_denote.png)
_and_hopping_(n_48)_assessments_for_r.jpeg)
._arrows_denote.png)
_and_hopping_(n_48)_assessments_for_r.jpeg)