INTRODUCTION
Quadriceps muscle strain, particularly involving the rectus femoris, is a common muscle injury in high-intensity dynamic sports such as soccer, which frequently involves kicking, sprinting, and turning motions.1 The incidence of this injury is comparable to that of hamstring strains and is recognized as a significant issue for athletes because of the extended time required for return to play and high recurrence rate.2,3 In particular, female athletes have an increased risk of muscle injury, potentially due to hormonal fluctuations associated with the menstrual cycle.2,4,5 Comparative studies have further demonstrated significantly higher risks of muscle injury in female athletes than in their male counterparts.6 These findings emphasize the need to develop effective rehabilitation strategies to prevent and reduce the recurrence of rectus femoris strain injuries.
The rectus femoris is a biarticular muscle that crosses both the hip and knee joints and is structurally predisposed to injury during dynamic high-load movements. In addition to injury history, several other risk factors have been identified, including reduced muscle flexibility,7 deficits in eccentric muscle strength, and bilateral strength asymmetry.8 Decreased muscle flexibility has also been associated with impaired skeletal muscle performance, such as reductions in peak torque and alterations in the angle at which the torque is produced.9,10 Thus, improving muscle flexibility may be key for both injury prevention and reducing recurrence.
Current rehabilitation strategies for muscle strain emphasize stretching and strengthening exercises in the subacute and later phases, with eccentric training being particularly effective in optimizing muscle length and preventing re-injury.11 However, residual scar tissue formation and joint motion range limitations often persist after strain injuries,10,12 leading to compromised flexibility and muscle function. Clinically, thermotherapy, including hot packs (HPs), high-frequency therapies, and ultrasound treatments, are widely used as an adjunct to address soft tissue impairments.13,14 Deep heating modalities can enhance soft tissue flexibility and improve local blood flow with greater physiological effects than superficial modalities such as HPs.15 Nevertheless, traditional high-frequency therapies have certain limitations, such as a narrow treatment area with ultrasound and thermal injury risk owing to the potential of localized overheating with conventional diathermy.13
Thus, 448-kHz capacitive resistive monopolar radiofrequency (CRMF) therapy has recently gained attention as a novel deep-heating modality. CRMF is a type of diathermy that generates uniform heat in deeper tissues. Although the depth of heating depends on the electrical resistance of the target area,16,17 CRMF can affect deeper structures such as tendons, ligaments, bones and cartilage.18 In addition, previous research has suggested that CRMF may affect a broader tissue area than therapeutic ultrasound, which may make it more suitable for treating large muscles such as the thigh muscles.19 Owing to its lower frequency than the conventional high-frequency devices, CRMF results in less superficial heating and is considered safer. Previous studies have reported that CRMF improves muscle extensibility and local tissue oxygen saturation in the trunk region, suggesting beneficial effects on muscle function and local hemodynamics.16,17 Nevertheless, its effects on the lower extremities, particularly the rectus femoris, which is a common site of strain injury, remain unclear.
Therefore, the present study examined the immediate effects of 448-kHz CRMF on muscle stiffness and hemodynamics of the rectus femoris in female collegiate soccer players. By comparing CRMF with HP therapy and a sham condition, the authors sought to clarify the physiological effects of each intervention and establish evidence for more effective rehabilitation approaches. It was hypothesized that CRMF would significantly reduce muscle stiffness and promote intramuscular blood flow compared with other intervention conditions.
METHODS
Participants
Twenty healthy female collegiate soccer players from a university team affiliated with the Kanto University Women’s Soccer League were recruited in July 2024. Rectus femoris strains are particularly prevalent among female soccer players, and sex-related differences in thermoregulatory responses may influence the physiological effects of thermal modalities; thus, this study only included women soccer players. Eligibility criteria included regular participation in team training and official matches for at least one year, regardless of playing position or competition level.
The exclusion criteria were as follows: (1) a history of thigh muscle injury or orthopedic disorders such as fractures within the previous year, (2) any history of neurological or cardiac disease, and (3) presence of a cardiac pacemaker. Participants who sustained new injuries during the study period or were unable to complete the required assessments were excluded from the analyses.
Study Design
This randomized crossover trial was conducted from August to October 2024 in a controlled athletic training facility at Juntendo University. The room temperature and humidity were maintained at 22–24 °C and 50%, respectively. Each participant received three interventions (CRMF, HP, and sham) on separate days, with the order of the conditions randomized using computer-generated randomization. The first intervention was randomly assigned; the second was randomly selected from the remaining two; and the third was the final unassigned condition. Because CRMF effects are typically reported to persist for approximately 30–45 min17,20,21 and longer-lasting effects have not been examined, a minimum two-day washout between sessions was adopted, consistent with previous protocols, to minimize potential carryover effects.20,21
Randomization and Blinding
The allocation sequence was generated using block randomization with computer software. Blinding was achieved by ensuring that participants were unaware of whether they received the CRMF or sham intervention. Owing to the perceptible heat stimulus, blinding was not feasible for the HP condition. Outcome measurements were performed by the same examiner administering the interventions, who was not blinded to the intervention condition. No modifications were made to the protocol after the trial commenced.
Interventions
All interventions were applied along the anatomical course of the rectus femoris from the anterior inferior iliac spine to the superior margin of the patella by a 5-year experienced (Master level) physiotherapist.
CRMF Intervention
The CRMF intervention was delivered using the INDIBA® Activ HCR801 (INDIBA, Barcelona, Spain) at a frequency of 448 kHz. This device includes capacitive (CAP) and resistive (RES) modes for superficial and deep heating, respectively. A 200 × 260 mm metal plate was placed on the posterior thigh as a passive electrode, with participants in the supine position. The output intensity was initially set at 50 W for the CAP mode and 30 W for the RES mode and increased by 1 W every 30 s until the participant reported a perceived thermal level of 6–7 on a 10-point scale. The total treatment time was 15 minutes (CAP 5 min; RES 10 min), and conductive gel was applied during treatment.
HP Intervention
A HP (PHYSIOPACK PPS-01, SAKAI Medical Co., Tokyo, Japan) was heated to 80 °C, wrapped in towels, and placed on the rectus femoris for 15 min. To standardize thermal sensation across modalities, perceived heat intensity during the HP intervention was assessed using the same 10-point thermal sensation scale as for CRMF, and towel layers were adjusted as needed to achieve a target rating of 6–7.
Sham Intervention
The sham intervention mimicked the CRMF procedure, and the participants were informed that they were receiving CRMF treatment. However, no current was applied during the 15-min session.
Outcome Measures
Muscle Stiffness (Tissue Elastic Modulus)
Muscle stiffness was assessed using shear wave elastography (SWE) (Aplio i300, Canon, Tochigi, Japan). The ultrasound probe was set to a frequency of 10 MHz, scanning depth of 1.5 cm, and field of view of 44 rps. Measurements were obtained at 66% of the distance from the patella to the anterior inferior iliac spine. The probe was applied mediolaterally with minimal compression (Supplemental Figure 1), and skin markings were used for consistent placement. Measurements were conducted in two postures: (1) resting (hip and knee at 0° flexion) and (2) stretching (measurement leg: hip 0°, knee 90°; non-measurement leg: hip 60°, knee 70°), using straps to secure the positions (Supplemental Figure 2). Before each assessment, participants were instructed to fully relax their thigh muscles and avoid voluntary contraction. The examiner visually monitored the thigh to confirm the absence of muscle contraction during measurement. For the analysis, the mean of three circular regions of interest (5 mm diameter) placed within a 10 × 10 mm area was used. Stiffness (kPa) was calculated using the formula elasticity = ρ × V², where ρ is the tissue density, and V is the shear wave velocity. To confirm measurement reliability, intra-session reliability was evaluated in five participants, yielding intraclass correlation coefficient ICC(1,3) = 0.989 in the resting position and ICC(1,3) = 0.899 in the stretching position.
Hemodynamics
Local hemodynamic parameters were assessed using a near-infrared spectroscopy device (BOM-L1TR, OMEGAWAVE, Tokyo, Japan), measuring oxygenated hemoglobin (Oxy-Hb), deoxygenated hemoglobin (deOxy-Hb), total hemoglobin (total-Hb), and tissue oxygen saturation (StO₂). The sensor was placed at the same site as the SWE probe, with emitter-detector distances set at 1.5 cm (superficial) and 3.0 cm (deep) to evaluate depth-dependent responses (Supplemental Figure 3). Test-retest reliability of NIRS-derived indices has been reported to be moderate to high for thigh muscle measurements.22
Measurement Protocol
To minimize the effects of sports activity, each session was generally conducted the day after a rest day. Participants were instructed to refrain from caffeine on the test day. Before baseline measurements, participants rested quietly in the supine position for 10 min to stabilize hemodynamic variables. Muscle stiffness and hemodynamics were assessed at three time points: pre-intervention, immediately post-intervention, and 30 min post-intervention. The measurement sequence was: (1) stiffness in the resting position, (2) hemodynamics in the resting position, and (3) stiffness in the stretching position. The participants remained in the supine position to minimize external variability.
Statistical Analyses
All statistical analyses were performed using SPSS version 29 (IBM Corp., Armonk, NY, USA). Normality of the primary outcome variables was assessed using the Shapiro–Wilk test. The change rates from baseline (pre-intervention) to post-intervention and 30 min post-intervention were calculated for each variable. A two-way repeated-measures analysis of variance (ANOVA) (3 conditions × 3 time points) was used to analyze differences, followed by Bonferroni-corrected post-hoc comparisons where appropriate. Sphericity was assessed using Mauchly’s test; when violated, Greenhouse–Geisser corrections were applied. The significance level was set at p < 0.05. Because an a priori sample size calculation was not performed before recruitment, post hoc power analysis was conducted using G*Power 3.1 software to estimate study power and Type II error likelihood.23 Additionally, effect sizes and 95% confidence intervals (CIs) were reported to indicate effect magnitude and precision. For pairwise comparisons, Cohen’s d was interpreted as small (0.20), medium (0.50), and large (0.80), whereas for ANOVA, partial eta squared (ηp²) was interpreted as small (0.01), medium (0.06), and large (0.14).24
Ethical Considerations
This study was approved by the Ethics Committee for Human Experiments of Juntendo University (No. 2024-24). All participants received detailed explanations of the study objectives and procedures and provided written informed consent prior to participation.
RESULTS
Participants
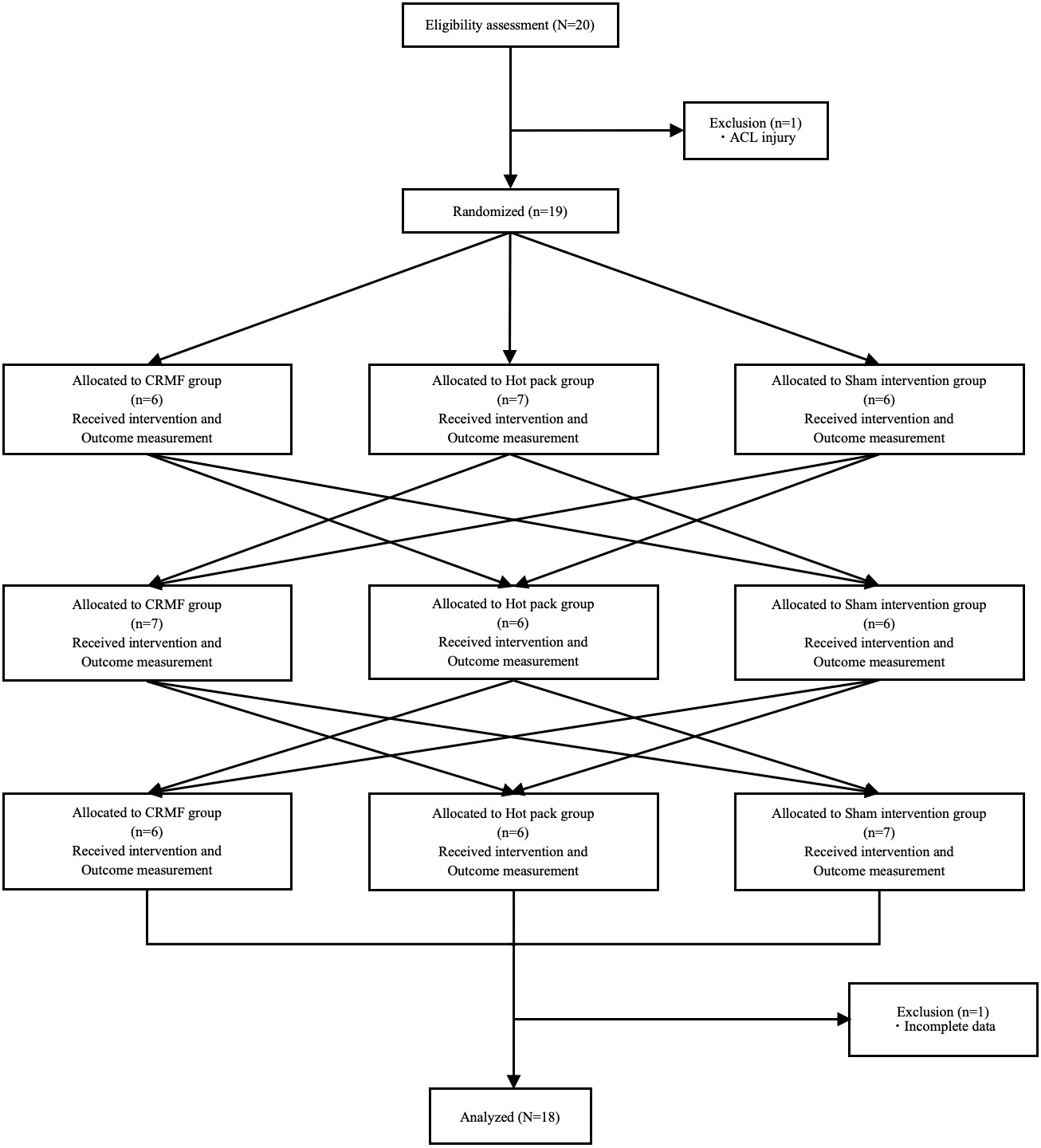
Of the 20 participants initially recruited, one was excluded due to an anterior cruciate ligament injury sustained prior to the first measurement, and another was excluded for failing to complete the full protocol due to injury. Thus, data from 18 participants (90%) were included in the final analyses (Figure 1). The characteristics of the final study cohort are listed in Table 1.
No adverse reactions related to HPs and CRMF interventions were reported by the participants. Absolute values for each outcome are shown in Supplemental Table 1.
Effects on Muscle Stiffness
In the stretched position, a significant interaction between condition and time (F = 4.838, p = 0.019, ηp² = 0.222, power = 0.940) was observed. The post-hoc tests showed that muscle stiffness immediately after CRMF (mean = 90.7) was significantly lower than that after HP (mean = 99.8, mean difference = −9.1, 95% CI [−14.2, −4.1], p < 0.001, Cohen’s d = 1.13) and sham interventions (mean = 102.5, mean difference = −11.8, 95% CI [−20.4, −3.1], p = 0.006, Cohen’s d = 0.85). However, no significant differences were observed among groups at 30 min post-intervention (CRMF vs HP: p = 0.583, CRMF vs sham: p = 1.000, HP vs sham: p = 0.432) (Table 2).
In the resting position, no significant condition-by-time interaction (F = 1.539, p = 0.201, ηp² = 0.083, power = 0.353) was observed, and neither the main effect of time (F = 0.646, p = 0.531, ηp² = 0.037) nor that of condition was significant (F = 2.993, p = 0.064, ηp² = 0.150) (Table 2, Supplemental Figure 4).
Hemodynamic Responses
Oxy-Hb
At 1.5-cm depth, no significant condition-by-time interaction (F = 2.078, p = 0.093, ηp² = 0.109, power = 0.589) and no significant main effect of time (F = 1.156, p = 0.327, ηp² = 0.064) were observed, whereas a significant main effect of condition was observed (F = 3.821, p = 0.032, ηp² = 0.184).
At a depth of 3 cm, a significant condition-by-time interaction (F = 5.616, p < 0.001, ηp² = 0.248, power = 0.971) was observed. Post hoc tests indicated that immediately post-intervention, Oxy-Hb was significantly higher in the CRMF and HP groups (means = 104.2 and 101.3, respectively) than in the sham group (mean = 95.2) (CRMF vs sham: mean difference = 9.1, 95% CI [2.2, 15.9], p = 0.008, Cohen’s d = 0.83, HP vs sham: mean difference = 6.1, 95% CI [0.7, 11.6], p = 0.025, Cohen’s d = 0.71), with no significant difference between the CRMF and HP groups (mean difference = 2.9, 95% CI [−3.1, 9.0], p = 0.643, Cohen’s d = 0.30). At 30 min post-intervention, Oxy-Hb remained significantly higher in the CRMF group (mean = 100.7) than in the sham group (mean difference = 6.5, 95% CI [1.9, 11.0], p = 0.005, Cohen’s d = 0.89). Nevertheless, no significant differences were observed in the other comparisons (CRMF vs HP: p = 0.201, HP vs sham: p = 0.552) (Supplemental Table 2, Figure 2).
deOxy-Hb
At a depth of 1.5 cm, no significant condition-by-time interaction (F = 1.963, p = 0.110, ηp² = 0.104, power = 0.562) and no significant main effects of time (F = 0.065, p = 0.937, ηp² = 0.004) or condition (F = 0.638, p = 0.534, ηp² = 0.036) were observed (Supplemental Table 3).
At a depth of 3 cm, a significant condition-by-time interaction (F = 2.691, p = 0.038, ηp² = 0.137, power = 0.718) was observed. Post hoc tests exhibited no significant between-group differences at any time point. Conversely, within the HP group, a significant change was observed from immediately post-intervention to 30 min post-intervention (p = 0.047).
Total-Hb
At a depth of 1.5 cm, no significant interaction between condition and time (F = 1.745, p = 0.150, ηp² = 0.093, power = 0.507) and no significant main effects of time (F = 0.679, p = 0.514, ηp² = 0.038) or condition (F = 1.843, p = 0.174, ηp² = 0.098) were observed (Supplemental Table 4, Figure 3).
At a depth of 3.0 cm, a significant condition-by-time interaction (F = 3.268, p = 0.016, ηp² = 0.161, power = 0.810) was observed. Post hoc tests demonstrated that, immediately after the intervention, total hemoglobin was significantly higher in the CRMF group (mean = 102.6) than in the sham group (mean = 96.1, mean difference = 6.5, 95% CI [0.6, 12.3], p = 0.028, Cohen’s d = 0.69). Nevertheless, no significant differences were observed between the CRMF and HP groups (p = 1.000) or between the HP and sham groups (p = 0.167), and no significant between-group differences were observed 30 min post-intervention (CRMF vs HP: p = 0.740, CRMF vs sham: p = 0.276, HP vs sham: p = 1.000).
StO₂
At 1.5 cm, a significant condition-by-time interaction (F = 3.284, p = 0.016, ηp² = 0.162, power = 0.812) was observed. Post hoc tests demonstrated that immediately after the intervention, StO₂ was significantly lower in the HP group (mean = 99.1) than in the sham group (mean = 102.1, mean difference = −3.0, 95% CI [−6.0, −0.1], p = 0.044, Cohen’s d = 0.64). Conversely, no significant differences were observed for CRMF vs HP (p = 0.066) or CRMF vs sham (p = 1.000). At 30 min post-intervention, no significant between-group differences were observed (CRMF vs HP: p = 0.418, CRMF vs sham: p = 0.167, HP vs sham: p = 1.000) (Supplemental Table 5, Figure 4).
At 3 cm, a significant condition-by-time interaction (F = 4.151, p = 0.005, ηp² = 0.196, power = 0.902) was observed. Post hoc tests indicated that immediately post-intervention, StO₂ was significantly higher in the CRMF group (mean = 101.8) than in the HP group (mean = 99.2, mean difference = 2.7, 95% CI [0.9, 4.4], p = 0.002, Cohen’s d = 0.97), whereas no significant differences were observed between CRMF and sham (p = 0.284) or between HP and sham (p = 0.745). At 30 min post-intervention, StO₂ was significantly higher in the CRMF group (mean = 102.4) than in the sham group (mean = 99.2, mean difference = 3.2, 95% CI [0.3, 6.1], p = 0.030, Cohen’s d = 0.68), with no significant differences between CRMF and HP (p = 0.250) or between HP and sham (p = 0.912).
DISCUSSION
This study investigated the immediate effects of 448-kHz CRMF on rectus femoris muscle stiffness and hemodynamics. CRMF significantly reduced muscle stiffness in the stretched position immediately post-intervention, outperforming both the HP and sham conditions. The magnitude of this reduction was large, suggesting a marked acute change. Furthermore, Oxy-Hb, total-Hb, and StO₂ were significantly elevated in the deeper tissue layers after CRMF, with some effects persisting for up to 30 min and exhibiting overall medium to large effect sizes. deOxy-Hb exhibited a significant condition × time interaction, although no significant between-group differences were observed at any time point. Together, these results suggest that CRMF reduces muscle stiffness and promotes local perfusion, particularly in deep muscle tissue, supporting the selection of certain thermal modalities to target deep muscle.
Changes in Muscle Stiffness
Only CRMF significantly reduced rectus femoris stiffness in the stretched condition immediately post-intervention, a result that was not observed in the HP or sham groups. Given the large effect size for this between-condition difference, the immediate muscle stiffness indicated a distinct physiological effect of CRMF. The primary mechanism of thermotherapy involves elevation of tissue temperature, leading to increased blood flow, muscle relaxation, and enhanced tissue elasticity. However, superficial heating modalities such as HP are limited in their ability to deliver effective thermal energy, penetrating depths ranging from less than 1cm up to 3 cm. This limitation is particularly evident in individuals with higher subcutaneous fat thickness, which is common among females and impedes deeper heat transmission.25–28
In contrast, CRMF utilizes Joule heating based on tissue-specific electrical resistance, enabling efficient deep-tissue heating independent of subcutaneous fat conductivity.17,25–29 The results of this study confirmed that HP produced no significant change in stiffness, likely because of its superficial thermal effects and variability in fat thickness across individuals.17,25,27 These findings are consistent with previous research showing reduced paraspinal muscle stiffness in the stretched condition after CRMF21 and extend its applicability to the rectus femoris. Collectively, these findings suggest that CRMF can acutely reduce stiffness in more than one muscle and raise the possibility that its effects may not be strictly site-specific. However, because only one thigh muscle was examined and tissue temperature at depth was not directly quantified, generalizability to other muscles and anatomical sites should be investigated in future studies.
Importantly, although stiffness reductions were evident immediately after CRMF, these effects diminished within 30 minutes, suggesting a transient physiological modulation. Previous studies have demonstrated that combining thermotherapy with stretching can enhance stretching effects.30 Therefore, when CRMF is combined with exercise therapy, such as stretching, stretching may be best performed immediately after CRMF. Further investigation is needed to determine optimal intervention combinations and timing.
Hemodynamic Responses
CRMF and HP both significantly increased Oxy-Hb levels in deep tissue (3 cm) immediately post-intervention compared with the sham group. Given the comparable effect sizes, the magnitude of the immediate Oxy-Hb increase may be similar for CRMF and HP. However, only CRMF maintained this elevation at 30 minutes, suggesting a sustained improvement in deep muscle perfusion. This contrast may be explained by differing thermal transmission mechanisms. HP relies on superficial conductive heating and may induce transient vasodilation via skin temperature elevation, whereas CRMF is thought to generate internal heat directly within deeper tissues, promoting stable and prolonged vasodilatory responses.29
Because deOxy-Hb showed no significant between-group differences at any time point, the lack of a clear deOxy-Hb rise alongside increased Oxy-Hb indicates that the effect likely stemmed from increased oxygen delivery rather than elevated metabolic demand, highlighting improved muscle oxygenation via enhanced arterial inflow.31 Increases in the total-Hb further support the hypothesis of local blood volume augmentation under CRMF, strengthening the evidence for circulatory enhancement.
These vascular effects may involve the modulation of autonomic function, particularly through attenuation of sympathetic tone and enhancement of parasympathetic activity, leading to peripheral vasodilation and increased muscle perfusion.17,31 The ability of CRMF to elicit deep, sustained blood flow enhancement distinguishes it from conventional thermotherapy, suggesting potential utility in interventions aimed at improving deep-tissue circulation.
Limitations
This study had some limitations. First, the study included only healthy young women soccer players, limiting the generalizability to other populations. Second, although measurements were scheduled the day after an off-training day and participants were asked to refrain from caffeine intake to reduce confounding, menstrual cycle phase and hydration status were neither standardized nor recorded; therefore, their potential influence on SWE and NIRS outcomes cannot be excluded. Third, participant blinding was not feasible for the HP condition, and the outcome assessor was not blinded to intervention allocation, potentially introducing expectancy and measurement bias. Fourth, skin and intramuscular temperatures were not measured; thus, interpretations regarding deep heating remain indirect. Additionally, application pressure was not quantified and may have differed between CRMF (including sham) and HP, potentially influencing superficial NIRS signals and partly explaining the lower StO₂ values observed in HP relative to sham at 1.5 cm depth immediately after the intervention. Fifth, subcutaneous tissue thickness was not measured, despite its potential to affect heat transmission and interpretation of both SWE and NIRS responses. Finally, only physiological markers were assessed, and functional outcomes, such as strength, fatigue resistance, or clinical symptoms (pain and range of motion) were not evaluated. Given that physiological improvements do not necessarily translate into functional recovery or injury prevention, future studies should incorporate performance-based and clinical endpoints and include direct temperature measurements and assessments of subcutaneous tissue thickness to clarify mechanisms and clinical relevance of CRMF.
CONCLUSION
The results of this study demonstrate that 448-kHz CRMF significantly reduced rectus femoris muscle stiffness in the stretched position and increased deep-tissue Oxy-Hb, total-Hb, and StO₂ in female collegiate soccer players. These effects were immediate, with some hemodynamic responses persisting for up to 30 minutes and were greater than those observed under conventional HP and sham conditions. Therefore, CRMF may represent an effective physical modality for short-term improvement of muscle stiffness and deep-tissue oxygenation and may be considered in selecting thermal modalities in sports and rehabilitation settings.
Acknowledgments
We would like to thank Editage (www.editage.com) for English language editing.
Funding
This study was supported by the Research Project of the Institute of Health and Sports Science & Medicine at Juntendo University.
Conflicts of Interest
None.
_across_conditions_analyzed_using_a_two-way.jpeg)
_across_conditions_analyzed_using_a_two-way_repe.jpeg)
_across_conditions_analyzed_using_a_two-.jpeg)
_across_conditions_analyzed_using_a_two-way.jpeg)
_across_conditions_analyzed_using_a_two-way_repe.jpeg)
_across_conditions_analyzed_using_a_two-.jpeg)